How Exercise Targets Atherosclerosis: From Prevention to Plaque Stabilization and Vascular Repair
Unlock the molecular benefits of exercise for heart disease prevention, including myokines, shear stress, and microRNA-driven vascular repair
EXERCISEHEART
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/9/202614 min read
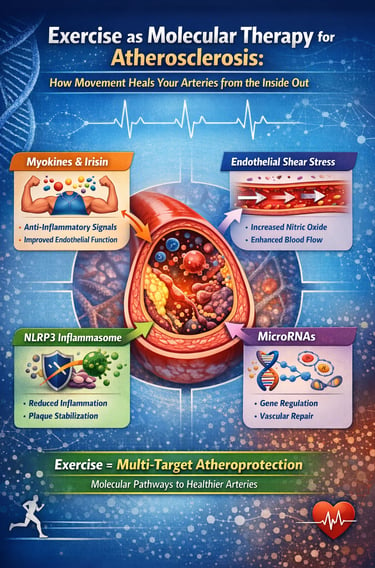
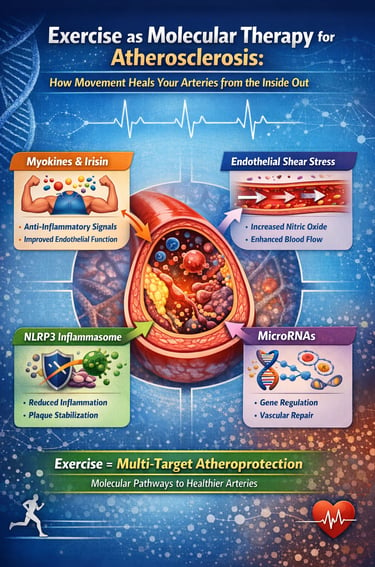
Exercise is not just prevention—it is molecular therapy for atherosclerosis.
Every workout triggers a cascade of protective signals inside your arteries, from anti-inflammatory myokines and improved endothelial function to suppression of plaque-destabilising pathways like the NLRP3 inflammasome. It also reshapes gene expression through microRNAs and reduces the burden of atherogenic ApoB-containing lipoproteins.
“Exercise reduces MACE risk by ~20–30% in longitudinal cohorts”
“VO₂ max improvement of 1 MET → ~10–15% mortality reduction”
“CRP reductions of ~20–40% with regular training”
Clinician’s Perspective
Exercise is not lifestyle advice—it is vascular pharmacology.
In modern cardiometabolic medicine, physical activity should be prescribed with the same intentionality as statins or antihypertensives. The difference is that exercise acts upstream, targeting the biology of disease initiation, not just downstream risk markers.Atherosclerosis is fundamentally inflammatory, and exercise is a multi-pathway anti-inflammatory therapy.
Beyond lipid-lowering, regular physical activity suppresses chronic low-grade inflammation through coordinated effects on cytokines, oxidative stress, and immune cell signalling. The inhibition of pathways such as the NLRP3 inflammasome represents a level of disease modification that most drugs only partially achieve.Endothelial function is the earliest and most reversible target.
Clinically, improvements in endothelial nitric oxide bioavailability occur within weeks of initiating aerobic exercise. This translates into better vasodilation, reduced arterial stiffness, and early plaque stabilization—well before structural changes are visible on imaging.ApoB particle burden—not just LDL-C—must frame our understanding.
Exercise favorably alters atherogenic lipoprotein dynamics by reducing ApoB-containing particle number and improving clearance. This is critical in patients with residual risk despite “normal” cholesterol levels.Skeletal muscle is an endocrine organ with systemic therapeutic reach.
Myokines such as irisin and exercise-induced IL-6 create an anti-inflammatory, metabolically favorable internal environment—linking muscle activity directly to vascular protection.Consistency outweighs intensity in long-term outcomes.
While high-intensity protocols amplify molecular responses, it is the chronic repetition of these signals—AMPK activation, mitochondrial adaptation, epigenetic modulation—that drives durable cardiovascular benefit.Exercise is most powerful when integrated, not isolated.
Combined aerobic and resistance training produces complementary effects across vascular, metabolic, and musculoskeletal systems—mirroring the multifactorial nature of atherosclerosis itself.The clinical shift is clear:
Exercise should move from being recommended to being prescribed, monitored, and followed up—as a first-line, disease-modifying therapy in cardiovascular care.
How Movement Heals Your Arteries From the Inside Out
If exercise were a drug, it would be the most powerful—and most underprescribed—therapy in cardiovascular medicine. It lowers the risk of heart attack, stabilises arterial plaques, reduces systemic inflammation, and improves survival across populations. Yet unlike conventional pharmacotherapy, its effects extend far beyond a single pathway. Exercise acts simultaneously on metabolism, vascular biology, immune signalling, and even gene expression—functioning not as a single agent, but as a coordinated, multi-target molecular intervention.
Atherosclerosis, the underlying cause of most cardiovascular events, is no longer viewed as a simple cholesterol storage disease. It is a dynamic, inflammatory condition driven by endothelial dysfunction, immune activation, lipid infiltration, and maladaptive cellular signalling. Traditional therapies—statins, antihypertensives, antiplatelets—target specific aspects of this process. Exercise, in contrast, engages the entire system. It improves endothelial nitric oxide bioavailability, suppresses pro-inflammatory pathways, enhances lipid handling, and promotes vascular repair mechanisms at the cellular level (Yin & Guo, 2025).
Recent advances in molecular physiology have revealed that contracting skeletal muscle behaves as an endocrine organ, releasing signalling molecules known as myokines that directly influence vascular health. At the same time, increased blood flow during exercise generates protective endothelial shear stress, while intracellular pathways such as AMP-activated protein kinase (AMPK) and the NLRP3 inflammasome are modulated in ways that reduce plaque progression and instability (Liu et al., 2026). Even more striking, exercise alters the expression of microRNAs—epigenetic regulators that reprogram vascular gene activity toward an anti-atherogenic profile (Ke et al., 2025).
In this light, exercise is not merely preventive—it is therapeutic at the deepest biological level. The arteries are not passive conduits; they are responsive, adaptive tissues. And with every bout of movement, they receive signals that can either accelerate disease or begin to reverse its course.
What Is Atherosclerosis? A Quick Primer
Atherosclerosis begins with damage to the inner lining of blood vessels — a single-cell-thick layer called the endothelium. When this lining is injured by high cholesterol, high blood pressure, smoking, or chronic inflammation, low-density lipoprotein (LDL) cholesterol infiltrates the arterial wall. Immune cells rush in, foam cells form, and over years, a fibrous plaque develops. As plaques grow, they narrow the artery; if they rupture, the result is a blood clot — and potentially a heart attack or stroke.
The disease is silent for decades. By the time symptoms appear, significant damage has already accumulated. This is why prevention and early biological intervention — including regular physical activity — matter so profoundly.
The Molecular Machinery: How Exercise Talks to Your Arteries
1. Myokines: Muscles as Medicine-Makers
When you exercise, your skeletal muscles do not merely contract and burn fuel — they become endocrine organs, secreting a class of signalling proteins called myokines directly into the bloodstream. These molecules travel to distant tissues including the heart, liver, and blood vessels, triggering protective biological cascades.
Two myokines have attracted particular scientific attention in the context of atherosclerosis: irisin and interleukin-6 (IL-6).
Irisin: The Vascular Guardian
Irisin is cleaved from a membrane protein called FNDC5 during aerobic exercise and released into the circulation. A landmark study published in Scientific Reports in 2026 by Inoue, Fujie, Uchida, and colleagues investigated the direct vascular effects of irisin in ApoE-deficient mice — a well-established animal model of atherosclerosis. Their findings were compelling: aerobic exercise-induced irisin secretion was significantly associated with improved endothelial function and a measurable reduction in atherosclerotic lesion development (Inoue et al., 2026). The study provided direct experimental evidence linking exercise-induced irisin to atheroprotection, strengthening the case for aerobic training as a first-line vascular therapy.
The mechanisms by which irisin protects the vasculature are multifaceted. Irisin appears to reduce oxidative stress within vessel walls, suppress inflammatory signalling pathways in endothelial cells, and promote nitric oxide (NO) bioavailability — a key molecule that keeps blood vessels relaxed and plaque-resistant.
Interleukin-6: The Anti-Inflammatory Paradox
IL-6 has a complicated reputation. When chronically elevated — as in metabolic syndrome or obesity — it is associated with systemic inflammation and increased cardiovascular risk. But exercise-derived IL-6 tells a very different story.
During acute aerobic or resistance exercise, working muscles release IL-6 in pulses that are transient, context-dependent, and fundamentally anti-inflammatory in character. This exercise-specific IL-6 response stimulates the production of anti-inflammatory cytokines such as IL-10 and IL-1 receptor antagonist (IL-1Ra), blunting the low-grade systemic inflammation that accelerates plaque formation (Yin & Guo, 2025).
A comprehensive systematic review with meta-analysis by Bettariga et al (2024), published in the Journal of Sport and Health Science, examined how different exercise training modes — aerobic training, resistance training, and high-intensity interval training (HIIT) — affect myokine expression in healthy adults. Their analysis revealed that exercise modality significantly influences the myokine secretion profile. Aerobic training was particularly effective at eliciting sustained irisin and IL-6 responses, while resistance training produced distinct myokine signatures with complementary benefits. This finding has immediate practical relevance: matching your exercise type to your cardiovascular goals is a meaningful clinical consideration.
2. Endothelial Shear Stress: The Mechanical Signal That Protects Vessels
Physical forces matter as much as chemical ones in vascular biology. When blood flows through an artery, it exerts a frictional drag force on the endothelial surface — a phenomenon called shear stress. Healthy, laminar (smooth) blood flow generates a pattern of shear stress that is profoundly protective to the endothelium. Disturbed or turbulent flow — such as occurs at arterial bends and bifurcations where plaques preferentially form — generates low or oscillatory shear stress that promotes dysfunction.
Aerobic exercise raises cardiac output and blood flow velocity, thereby increasing laminar shear stress throughout the vasculature. This is not merely a passive mechanical effect. Endothelial cells are exquisitely sensitive to shear forces through mechanoreceptors and ion channels. When stimulated by exercise-induced shear, they upregulate endothelial nitric oxide synthase (eNOS), produce more nitric oxide, suppress adhesion molecule expression (which draws inflammatory cells into the vessel wall), and reduce oxidative stress (Yin & Guo, 2025).
In practical terms: every bout of sustained aerobic exercise is a training session not just for your muscles, but for your arteries. With repeated exposure, vessels become more responsive, more elastic, and more resistant to the inflammatory and lipid-driven insults that initiate plaque.
3. Silencing the NLRP3 Inflammasome: Exercise Turns Off a Key Inflammation Switch
One of the most exciting recent developments in cardiovascular molecular biology concerns the NLRP3 inflammasome — a multiprotein complex that acts as a danger sensor inside immune cells. When activated by oxidised LDL, cholesterol crystals, or metabolic stress — all features of atherosclerotic lesions — NLRP3 triggers the release of powerful pro-inflammatory cytokines (IL-1β and IL-18) that accelerate plaque instability and rupture.
A detailed review by Liu, Hu, Wang, and Ding (2026), published in Current Molecular Medicine, examined the growing body of evidence showing that exercise alleviates atherosclerosis in part by suppressing NLRP3 inflammasome activation. Exercise achieves this through multiple converging pathways:
Reduction of oxidative stress: Exercise training enhances antioxidant enzyme systems (superoxide dismutase, glutathione peroxidase), reducing the reactive oxygen species that serve as NLRP3 activators.
Autophagy promotion: Exercise stimulates the cellular housekeeping process of autophagy, which clears the damaged organelles and cholesterol crystals that would otherwise trigger NLRP3 assembly.
AMP-activated protein kinase (AMPK) activation: AMPK — a master energy-sensing enzyme activated during exercise — directly inhibits NLRP3 inflammasome priming, interrupting the inflammatory cascade before it begins.
Mitochondrial health: Regular exercise improves mitochondrial function and reduces mitochondrial-derived reactive oxygen species, removing a major driver of NLRP3 activation in macrophages within plaques (Liu et al., 2026).
This NLRP3 pathway represents one of the most mechanistically specific connections yet identified between physical activity and plaque stabilisation. Unstable plaques rupture and cause heart attacks; stable plaques, even if not dramatically smaller, are far less dangerous. Exercise, by suppressing NLRP3 activity, may literally prevent the final catastrophic event in the atherosclerosis story.
4. Exercise-Regulated MicroRNAs: Reprogramming Vascular Gene Expression
Among the most frontier-level discoveries in this field is the role of microRNAs (miRNAs) — small non-coding RNA molecules that fine-tune gene expression by preventing specific messenger RNAs from being translated into protein. Exercise induces changes in the circulating levels of numerous miRNAs, several of which have direct atheroprotective functions.
A thorough review by Ke, Cao, Lu, Xu, Zhang, Xu, and Jiang (2025), published in Atherosclerosis, outlined the emerging landscape of exercise-regulated miRNAs and their roles in vascular protection. Key findings include:
miR-126: Upregulated by exercise-induced shear stress, miR-126 promotes endothelial repair, supports vascular smooth muscle cell integrity, and suppresses expression of genes that drive monocyte recruitment into plaques.
miR-146a: This miRNA, elevated by exercise, acts as a brake on the NF-κB inflammatory pathway — a master regulator of vascular inflammation — thereby reducing adhesion molecule expression on the endothelial surface.
miR-21: Modulated by aerobic training, miR-21 plays complex roles in smooth muscle cell behaviour within plaques, influencing their phenotypic switching from a quiescent to a proliferative state — a critical step in lesion progression (Ke et al., 2025).
What makes the microRNA story particularly profound is its epigenetic dimension. These molecules can reprogram gene expression patterns that persist beyond any single exercise bout. Regular training may, in effect, reshape the transcriptional landscape of your vascular cells — making them inherently more resistant to the molecular events that drive atherosclerosis. This is biological memory, written not in DNA sequence, but in the regulatory instructions that govern how genes are read.
The Comprehensive Picture: Exercise as Multi-Target Vascular Therapy
Yin and Guo (2025), in their wide-ranging review published in Frontiers in Cell and Developmental Biology, synthesised the full scope of exercise's beneficial effects on atherosclerosis, integrating the myokine, shear stress, inflammasome, and metabolic dimensions into a coherent framework. Their work highlights that no single mechanism operates in isolation. Instead, exercise orchestrates a network of complementary protective effects:
Improved lipid profiles (lower LDL, higher HDL)
Reduced blood pressure and arterial stiffness
Enhanced endothelial nitric oxide production
Systemic reduction of inflammatory markers (CRP, TNF-α, IL-6 in its chronic form)
Improved insulin sensitivity and glucose regulation
Weight management, reducing adipose tissue-derived inflammatory signals
The convergence of all these effects explains why physical activity remains, by some metrics, more powerful than many pharmaceutical interventions in reducing cardiovascular mortality — particularly when implemented consistently over years.
Practical Applications: Translating Molecular Science Into Daily Movement
Understanding the mechanisms above is valuable because it allows us to exercise strategically, not just effortfully.
For irisin and myokine responses: Moderate-to-vigorous aerobic exercise (brisk walking, jogging, cycling, swimming) performed for 30–60 minutes on most days of the week generates the most robust irisin secretion. Intensity matters: higher-intensity intervals (HIIT) can amplify myokine responses even within shorter sessions.
For endothelial shear stress: Any exercise that sustainably elevates heart rate and blood flow is beneficial. Activities involving large muscle groups — running, rowing, cycling — are particularly effective. Even moderate-intensity walking (5,000–8,000 steps per day) meaningfully improves laminar shear stress patterns over time.
For NLRP3 suppression and inflammation reduction: Consistency is paramount. The AMPK and mitochondrial pathways that suppress NLRP3 require repeated activation. A regular weekly exercise rhythm — not occasional bursts — is the biological key. Both aerobic and resistance training contribute here.
For microRNA reprogramming: Evidence suggests that both aerobic and resistance training modulate different but complementary miRNA profiles. A mixed training programme — combining cardiovascular exercise with strength work — may produce the broadest epigenetic vascular benefits.
Practical weekly template:
Monday/Wednesday/Friday: 30–45 minutes moderate aerobic exercise (brisk walk, cycle, swim)
Tuesday/Thursday: 20–30 minutes resistance training (bodyweight, bands, or weights)
Saturday: 45–60 minutes recreational activity (hiking, sport, yoga)
Sunday: Rest or gentle movement (15–20-minute walk)
Before you begin: Always consult your doctor or a cardiac rehabilitation specialist before starting a new exercise programme — particularly if you have existing cardiovascular disease, recent cardiac events, or significant risk factors. Exercise is therapy, and like all therapy, it benefits from professional guidance.
Frequently Asked Questions
Q1: How quickly does exercise start protecting my arteries? Measurable improvements in endothelial function can be detected after as little as 4–8 weeks of consistent aerobic training. Some acute benefits — including improved blood flow and transient anti-inflammatory myokine release — occur with a single exercise bout. Long-term structural benefits, including plaque stabilisation and microRNA-driven gene reprogramming, develop over months to years of regular training.
Q2: Is aerobic exercise better than resistance (strength) training for atherosclerosis? Both are beneficial, and research suggests they work through partially different mechanisms. Aerobic training is particularly potent for irisin secretion, endothelial shear stress improvement, and NLRP3 suppression. Resistance training contributes complementary myokine profiles, metabolic benefits, and systemic inflammation reduction. The ideal programme combines both modalities, as supported by Bettariga et al. (2024).
Q3: Can exercise reverse existing atherosclerotic plaques? Current evidence suggests exercise can stabilise existing plaques and slow or halt their progression, but fully reversing established calcified plaques is less clearly demonstrated. The greatest value lies in stabilising vulnerable (rupture-prone) plaques and preventing new lesion development. Even in people with established disease, exercise significantly reduces the risk of cardiovascular events.
Q4: What is irisin and why does it matter for heart health? Irisin is a myokine — a protein secreted by working muscles during aerobic exercise. Research by Inoue et al. (2026) demonstrated that aerobic exercise-induced irisin secretion is directly associated with improved endothelial function and reduced atherosclerotic lesion formation. It represents one of the clearest molecular links between physical activity and cardiovascular protection.
Q5: I have cardiovascular disease — is exercise safe for me? For the vast majority of people with stable cardiovascular disease, supervised exercise is not only safe but strongly recommended by all major cardiology guidelines. Cardiac rehabilitation programmes specifically use structured exercise to improve outcomes after heart attacks and procedures. The key is working with your healthcare provider to determine appropriate intensity and monitoring.
Q6: How many steps per day do I need for vascular benefits? While 10,000 steps is a popular target, evidence suggests meaningful cardiovascular benefits begin accruing at 5,000–7,000 steps daily, with benefits plateauing somewhat above 8,000–10,000 steps in most populations. More important than hitting a specific target is creating a consistent daily movement habit.
Q7: Can diet and exercise together have an additive effect on atherosclerosis? Absolutely. A Mediterranean-style diet (rich in vegetables, olive oil, fish, nuts, and whole grains) and regular physical activity work through complementary and sometimes synergistic molecular pathways. Diet influences lipid profiles, gut microbiome composition, and systemic inflammation; exercise adds the shear stress, myokine, and microRNA dimensions. Together, they represent the most powerful non-pharmacological cardiovascular protection strategy available.
Clinical pearls
1. The Shear Stress "Vascular Polish"
Pulsatile laminar shear stress induced by aerobic exercise activates mechanotransducers on the endothelial surface. This triggers the phosphorylation of eNOS (endothelial Nitric Oxide Synthase), increasing NO bioavailability and suppressing the expression of VCAM-1 and ICAM-1 (adhesion molecules).
Think of blood flow during exercise as a "power wash" for your arteries. It physically signals the inner lining of your blood vessels to produce a natural gas (Nitric Oxide) that keeps the pipes slippery, relaxed, and resistant to the "sticky" cells that cause plaque.
2. Myokines: Muscle as an Endocrine Organ
Skeletal muscle contraction induces the expression of FNDC5, which is cleaved into the myokine Irisin. Irisin circulates to the vasculature where it reduces oxidative stress and promotes a "browning" of adipose tissue, improving the systemic cardiometabolic profile.
Every time you move, your muscles act like a pharmacy, manufacturing and releasing "health chemicals" (myokines) into your blood. These chemicals travel to your heart and arteries to help repair damage and reduce inflammation—benefits you can't get from a pill.
3. The IL-6 "Anti-Inflammatory Pulse"
Unlike the chronic, low-grade IL-6 elevation seen in obesity (which is pro-inflammatory), exercise-induced IL-6 pulses are transient and induce the release of IL-10 and IL-1Ra. This creates an anti-inflammatory environment that directly counteracts the systemic inflammation driving atherosclerosis.
There is "bad" inflammation (the kind that stays high and damages your heart) and "good" inflammation (the short spike you get during a workout). The short spike during exercise actually trains your body to shut down the bad inflammation, protecting your arteries from long-term damage.
4. Silencing the NLRP3 "Inflammation Switch"
Exercise suppresses the NLRP3 inflammasome by enhancing autophagy and reducing mitochondrial reactive oxygen species (mtROS). By inhibiting the maturation of IL-1β, exercise stabilizes the fibrous cap of existing plaques, reducing the risk of acute rupture.
Some plaques in your arteries are like "volcanoes"—unstable and prone to erupting into a heart attack. Exercise works like a cooling system that turns off the "fire" inside those plaques, making them much more stable and significantly less likely to cause a medical emergency.
5. MicroRNA Epigenetic Reprogramming
Regular physical activity modulates circulating microRNAs (e.g., miR-126 and miR-146a). These non-coding RNAs act as epigenetic "brakes" on pro-atherogenic gene expression, effectively reprogramming the vascular cells to a more resilient phenotype.
Exercise doesn't just burn calories; it actually talks to your genes. It tells your body to "turn off" the genes that build plaque and "turn on" the genes that repair your blood vessels. This "genetic reprogramming" means that the more consistent you are, the more your body naturally protects itself.
6. The AMPK Metabolic Master Switch
Exercise increases the AMP-to-ATP ratio, activating AMPK (AMP-activated protein kinase). This master regulator inhibits the mTOR pathway and activates PGC-1α, improving mitochondrial biogenesis and reducing the metabolic "fuel" that drives the formation of foam cells within the arterial wall.
Exercise turns on a master energy switch in your cells called AMPK. This switch forces your body to clean out cellular "trash" and become more efficient at burning fuel. This prevents the buildup of waste products in your artery walls that would otherwise turn into plaque.
Author’s Note
As a clinician, I have spent years treating the downstream consequences of atherosclerosis—heart attacks, heart failure, and progressive vascular disease. Yet one truth becomes clearer with time and evidence: many of these outcomes are not inevitable. They are, to a significant extent, biologically modifiable.
This article was written to shift the conversation from exercise as general advice to exercise as precise molecular therapy. The science presented here is drawn from contemporary research in vascular biology, immunology, and exercise physiology, but its implications are deeply practical. Every pathway discussed—myokines, endothelial shear stress, inflammasome regulation, and microRNA signaling—represents a lever that can be activated through something as accessible as regular physical movement.
At the same time, it is important to maintain clinical balance. Exercise is powerful, but it is not a replacement for evidence-based medical therapy where indicated. Rather, it is a foundational intervention—one that works synergistically with pharmacological treatments to reduce risk, improve function, and enhance long-term outcomes.
My goal is not only to inform but to reframe. When patients understand how exercise works at a biological level, adherence improves. When clinicians view it as a measurable, dose-dependent therapy, prescription becomes more intentional.
Ultimately, this is about reclaiming one of the most effective tools in medicine—one that is universally available, biologically potent, and too often underutilized.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Is Your Heart at Risk? How the TG/HDL Ratio Can Warn You Early | DR T S DIDWAL
Exercise and Vascular Health: How Nitric Oxide and Endothelial Function Keep Your Arteries Young
How Exercise Boosts Nitric Oxide and Blood Flow: The Endothelial Shear Stress Mechanism
References
Bettariga, F., Taaffe, D. R., Galvão, D. A., Lopez, P., Bishop, C., Markarian, A. M., Natalucci, V., Kim, J.-S., & Newton, R. U. (2024). Exercise training mode effects on myokine expression in healthy adults: A systematic review with meta-analysis. Journal of Sport and Health Science, 13(6), 764–779. https://doi.org/10.1016/j.jshs.2024.03.005
Inoue, K., Fujie, S., Uchida, M., et al. (2026). Aerobic exercise-induced irisin secretion is associated with improved endothelial function and reduced atherosclerosis in ApoE-deficient mice. Scientific Reports. https://doi.org/10.1038/s41598-026-39903-8
Ke, S., Cao, X., Lu, X., Xu, J., Zhang, C.-Y., Xu, L., & Jiang, X. (2025). Atheroprotective roles of exercise-regulated microRNAs. Atherosclerosis, 405, Article 119229. https://doi.org/10.1016/j.atherosclerosis.2025.119229
Liu, X., Hu, S., Wang, X., & Ding, Y. (2026). Exercise alleviates atherosclerosis through the modulation of the NLRP3 inflammasome. Current Molecular Medicine, 26(4), 358–375. https://doi.org/10.2174/0115665240368171250419113225
Yin, M. T., & Guo, L. (2025). Exercise in atherosclerosis: its beneficial effects and underlying mechanism. Frontiers in Cell and Developmental Biology, 13, 1598794. https://doi.org/10.3389/fcell.2025.1598794