Could High Insulin Be the Real Cause of Obesity and Diabetes?
Learn why chronically high insulin levels are linked to obesity, cardiovascular disease, inflammation, fatty liver, and cancer biology.
DIABETESOBESITY
Dr. T.S. Didwal, M.D.(Internal Medicine)
5/10/202612 min read
Key Takeaways
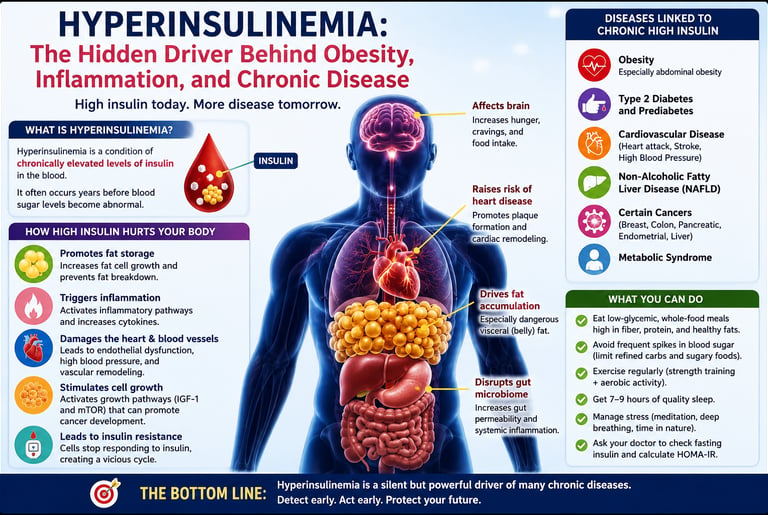
Hyperinsulinemia refers to chronically high insulin levels and may develop years before blood sugar becomes abnormal or diabetes is diagnosed.
Emerging research suggests high insulin may contribute to obesity, inflammation, cardiovascular disease, fatty liver disease, and certain cancers — not just result from them.
Standard glucose tests can miss early metabolic dysfunction, because fasting glucose and HbA1c may remain normal while insulin levels are already elevated.
Lifestyle interventions can significantly improve insulin sensitivity, including regular exercise, high-fiber nutrition, weight management, stress reduction, quality sleep, and reducing ultra-processed foods.
Early detection matters. Measuring fasting insulin, HOMA-IR, and other metabolic markers may help identify cardiometabolic risk before diabetes develops.
For decades, insulin has been viewed as a lifesaving hormone whose primary role is to regulate blood glucose. In many ways, that understanding is correct. Without insulin, life is impossible. But emerging metabolic research suggests that chronically elevated insulin — a condition known as hyperinsulinemia — may also be one of the earliest and most overlooked triggers of modern chronic disease (Guess et al., 2026). Long before blood sugar rises into the diabetic range, insulin levels can climb silently for years, reshaping metabolism in ways that promote fat storage, inflammation, cardiovascular dysfunction, and even cancer biology (Zhang et al., 2021).
Traditionally, medicine has treated hyperinsulinemia as a secondary consequence of obesity and insulin resistance. However, a growing body of evidence now challenges that sequence, suggesting that elevated insulin itself may actively drive weight gain and metabolic deterioration rather than simply respond to it (Guess et al., 2026; Chawla, 2023). Excess insulin suppresses fat breakdown, stimulates adipose tissue growth, alters hunger signaling in the brain, and activates inflammatory pathways throughout the body (Zhang et al., 2021). Over time, this creates a self-reinforcing metabolic cycle that increases the risk of type 2 diabetes, cardiovascular disease, fatty liver disease, and several obesity-associated cancers.
Perhaps most concerning is that hyperinsulinemia often remains clinically invisible. Standard screening tests, such as fasting glucose or HbA1c, may appear normal while metabolic damage is already underway (Bkaily et al., 2025). By the time diabetes is formally diagnosed, years of vascular, inflammatory, and hormonal dysfunction may have already occurred.
Understanding hyperinsulinemia may therefore represent one of the most important shifts in modern preventive medicine — moving healthcare from reactive glucose management to early metabolic intervention.
What Is Hyperinsulinemia, and Why Does It Matter?
Hyperinsulinemia refers to abnormally high levels of insulin circulating in the bloodstream. In a healthy body, insulin is released by the pancreatic beta cells in precise, short bursts in response to rising blood glucose. It acts as a key that unlocks cells and allows glucose to enter for energy.
In hyperinsulinemia, this finely tuned system becomes dysregulated. The pancreas produces excess insulin — either because of peripheral insulin resistance (cells stop responding normally) or, increasingly researchers believe, as a primary physiological defect that precedes resistance.
The consequences cascade rapidly:
Fat storage increases, and fat breakdown is suppressed
Chronic systemic inflammation is triggered
Cardiovascular remodeling begins
Cell growth pathways become hyperactivated
The risk of cancer, heart disease, and diabetes climbs significantly
What makes hyperinsulinemia particularly dangerous is that it is largely invisible in routine clinical practice. Standard fasting glucose tests can appear normal for years while insulin levels silently rise. This "quiet" metabolic disruption is now recognized as a central driver across multiple chronic disease pathways.
Hyperinsulinemia as a Cause of Obesity — Not Just a Result
Perhaps the most provocative and clinically important reframing comes from a landmark 2026 review published in Nature Reviews Endocrinology by Guess et al., who argue that hyperinsulinemia is causally upstream of obesity rather than simply downstream of it.
The conventional model holds that excess caloric intake creates fat mass, which promotes inflammation and insulin resistance, eventually leading to compensatory hyperinsulinemia. Guess et al. (2026) challenge this with compelling mechanistic and epidemiological evidence showing that elevated insulin itself:
Drives adipogenesis (fat cell creation and enlargement)
Suppresses lipolysis (the breakdown and burning of stored fat)
Promotes positive energy balance by altering hunger signaling in the hypothalamus
Creates a self-reinforcing feedback loop that perpetuates weight gain independently of caloric intake
This reframing has significant implications for public health. If insulin elevation precedes and promotes obesity, then interventions targeting insulin reduction — through dietary composition, meal timing, pharmacology, or lifestyle modification — may need to be prioritized far earlier in clinical care, well before frank obesity or diabetes develops.
Chawla (2023), writing in the International Journal of Diabetes in Developing Countries, reinforces this perspective by describing hyperinsulinemia, obesity, and T2D as part of a metabolic continuum rather than discrete, sequential disorders. In this continuum model, hyperinsulinemia occupies the earliest and most modifiable position — making it the most promising target for prevention.
Note on the Scientific Debate: While emerging research, including the 2026 review by Guess et al., suggests that hyperinsulinemia may act as an upstream driver of obesity and metabolic disease in many individuals, the scientific community continues to debate the exact sequence of events. Many experts view hyperinsulinemia and insulin resistance as bidirectional — each can reinforce the other. Regardless of which comes first, reducing chronically elevated insulin through lifestyle and medical interventions remains a promising and practical strategy for improving metabolic health.
The Inflammation Connection: How Excess Insulin Sets the Body on Fire
Chronic, low-grade systemic inflammation is now widely recognized as a pathological thread linking obesity, diabetes, cardiovascular disease, and certain cancers. What drives this inflammation in metabolically compromised individuals? Hyperinsulinemia is a central suspect.
Zhang et al. (2021), in their comprehensive review published in Diabetes & Metabolism Journal, detail how hyperinsulinemia promotes inflammation through several interconnected pathways:
1. Activation of Pro-Inflammatory Signaling Cascades
Excess insulin overstimulates the PI3K/Akt and MAPK/ERK signaling pathways. While these are normal components of cellular insulin signaling, chronic overstimulation promotes the transcription of pro-inflammatory cytokines, including tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6).
2. Adipose Tissue Dysfunction
In states of chronic hyperinsulinemia, adipose (fat) tissue undergoes structural and functional changes. Adipocytes become enlarged (hypertrophic), and the tissue becomes infiltrated by immune cells, particularly macrophages. This inflamed adipose tissue releases a stream of adipokines — bioactive molecules that perpetuate systemic inflammation far beyond the fat depot itself.
3. Gut Microbiome Disruption
Emerging evidence highlighted by Zhang et al. (2021) also implicates hyperinsulinemia in gut microbiome dysbiosis, which further amplifies intestinal permeability ("leaky gut") and allows bacterial lipopolysaccharides (LPS) into systemic circulation — potently triggering inflammatory responses.
Wiebe et al. (2026), in a prospective cohort study published in Diabetes, Obesity and Metabolism, provide some of the strongest population-level evidence yet. Their large-scale study directly examined the combined and independent contributions of obesity, systemic inflammation, and hyperinsulinemia to the incidence of non-communicable chronic diseases (NCDs) and all-cause mortality. Their findings confirm that:
Hyperinsulinemia, even when analyzed independently of BMI, significantly increases NCD incidence
The interaction between hyperinsulinemia and systemic inflammation multiplicatively amplifies disease risk
Mortality risk escalates substantially when all three factors — obesity, inflammation, and hyperinsulinemia — co-occur
This cohort evidence is crucial: it moves the conversation from mechanism to measurable human outcomes, showing that hyperinsulinemia is not an abstract laboratory phenomenon but a clinically significant and independently lethal condition.
Hyperinsulinemia and the Cardiovascular System: A Heart Under Siege
The cardiovascular system is one of the earliest and most severely affected targets of chronic hyperinsulinemia. Bkaily et al. (2025), in a detailed pathophysiological analysis published in Biomedicines, trace the molecular and cellular events through which prediabetic hyperinsulinemia and insulin resistance remodel the heart and vasculature.
Vascular Endothelial Dysfunction
Normally, insulin supports vascular health by stimulating nitric oxide (NO) production in endothelial cells, promoting vasodilation. In hyperinsulinemia, however, signaling becomes imbalanced. The PI3K pathway — responsible for NO synthesis — becomes blunted due to insulin resistance, while the MAPK pathway remains overactive. The result is a shift toward vasoconstriction, platelet aggregation, and endothelial inflammation, the foundational steps in atherosclerosis.
Cardiac Structural Remodeling
Bkaily et al. (2025) describe how excess insulin directly affects cardiomyocytes (heart muscle cells), altering calcium handling, promoting hypertrophic growth, and increasing oxidative stress. Over time, these cellular changes translate into left ventricular hypertrophy, diastolic dysfunction, and increased susceptibility to arrhythmia — long before overt diabetes is diagnosed.
The Prediabetic Window: A Critical and Overlooked Stage
One of the most clinically urgent insights from Bkaily et al. (2025) is that cardiovascular damage begins during prediabetes — the phase where fasting glucose is elevated but below the diabetic threshold, yet insulin levels are already chronically high. This means that millions of people currently classified as "not yet diabetic" may already be experiencing measurable cardiovascular injury.
This underscores the importance of measuring fasting insulin and insulin-to-glucose ratios as part of routine cardiovascular screening, rather than relying solely on HbA1c or fasting glucose.
Hyperinsulinemia and Cancer: Fueling Abnormal Cell Growth
Perhaps the most alarming frontier in hyperinsulinemia research is its connection to cancer biology. Zhang et al. (2021) dedicate considerable attention to the mechanisms by which chronically elevated insulin promotes tumorigenesis:
Insulin as a Growth Factor
Insulin shares structural similarity with insulin-like growth factor-1 (IGF-1) and can activate IGF-1 receptors on cells. Both insulin and IGF-1 receptors, when chronically stimulated, activate mTOR (mechanistic target of rapamycin) — one of the master regulators of cell growth, proliferation, and survival. This creates a biochemical environment permissive to tumor development and progression.
Hyperinsulinemia-Associated Cancers
Zhang et al. (2021) identify particularly strong links between hyperinsulinemia and:
Colorectal cancer
Breast cancer (especially in postmenopausal women)
Pancreatic cancer
Endometrial cancer
Liver cancer
The Insulin-Estrogen Axis
In women, hyperinsulinemia additionally suppresses sex hormone-binding globulin (SHBG), leading to elevated free estrogen levels. This estrogen excess is an established driver of breast and endometrial cancers, creating a direct endocrine link between metabolic dysfunction and gynecological malignancy.
Wiebe et al. (2026) corroborate these cancer-related risks at the population level, finding that individuals with concurrent obesity, inflammation, and hyperinsulinemia faced the highest cancer-related NCD burden in their cohort — reinforcing the clinical and public health urgency of addressing insulin dysregulation proactively.
Practical Applications: What You and Your Doctor Can Do Right Now
Understanding the science is powerful. But translating it into actionable steps is what changes lives. Here are evidence-informed strategies to reduce hyperinsulinemia and break the cycle:
1. Prioritize Low-Glycemic, High-Fiber Nutrition
Reducing the frequency and magnitude of insulin spikes is foundational. Diets built around:
Non-starchy vegetables (broccoli, leafy greens, cauliflower)
Legumes and whole grains (lentils, barley, oats)
Lean proteins and healthy fats (fish, nuts, olive oil)
...help reduce postprandial insulin surges. Consistent evidence supports low-glycemic and Mediterranean-style dietary patterns as particularly effective.
2. Consider Time-Restricted Eating (Intermittent Fasting)
Extending overnight fasting windows (e.g., 12–16 hours) allows insulin levels to fall and remain low for longer periods, giving cells time to restore insulin sensitivity. Even modest fasting periods (12 hours) have shown measurable benefits on fasting insulin levels.
3. Prioritize Both Aerobic and Resistance Exercise
Physical activity is one of the most potent insulin-sensitizing interventions available. Aerobic exercise clears blood glucose acutely, while resistance training builds muscle mass — the largest insulin-mediated glucose disposal organ in the body. Aim for at least 150 minutes of moderate activity per week, combined with 2–3 resistance sessions.
4. Optimize Sleep Quality and Duration
Chronic sleep deprivation elevates cortisol and disrupts insulin signaling significantly. Targeting 7–9 hours of quality sleep is a metabolic intervention, not just a lifestyle preference.
5. Ask for Fasting Insulin Testing
If you have risk factors (family history of diabetes, excess abdominal fat, fatigue, sugar cravings, PCOS, skin tags), ask your physician to measure:
Fasting serum insulin (target: <8–10 µIU/mL)
HOMA-IR (homeostasis model assessment of insulin resistance; target: <2.0)
Fasting insulin-to-glucose ratio
Early detection of rising insulin — before glucose becomes abnormal — allows for intervention during the most modifiable window.
6. Discuss Pharmacological Options if Appropriate
For individuals with established hyperinsulinemia and at-risk profiles, medications such as metformin, GLP-1 receptor agonists, and SGLT-2 inhibitors have demonstrated insulin-lowering and cardiometabolic-protective effects. These are clinical decisions to make collaboratively with your healthcare provider.
7. Manage Chronic Stress Actively
Psychological stress elevates cortisol, which directly raises blood glucose and compensatory insulin. Mindfulness-based stress reduction (MBSR), yoga, and cognitive behavioral strategies are not "soft" interventions — they have measurable effects on insulin and inflammatory biomarkers.
Frequently Asked Questions (FAQs)
❓ FAQ 1: Is hyperinsulinemia the same as diabetes?
No — hyperinsulinemia is not the same as diabetes, though the two conditions are closely related. Diabetes is characterized by elevated blood glucose, whereas hyperinsulinemia refers specifically to elevated blood insulin. In fact, hyperinsulinemia often precedes diabetes by years or decades. During this pre-diabetic window, the pancreas is working overtime to compensate for insulin resistance, producing excess insulin to keep blood sugar normal. Chawla (2023) describes this as part of a metabolic continuum, with hyperinsulinemia representing the earliest and most reversible stage.
❓ FAQ 2: Can you have hyperinsulinemia if you are not overweight?
Yes. While obesity and hyperinsulinemia are strongly linked, you do not need to be overweight to have elevated insulin. "Metabolically obese normal weight" (MONW) individuals — those with a healthy BMI but excess visceral fat and poor metabolic fitness — can have clinically significant hyperinsulinemia. Genetic factors, physical inactivity, poor dietary quality, and sleep disruption can all drive hyperinsulinemia independently of body weight.
❓ FAQ 3: How is hyperinsulinemia diagnosed?
Standard glucose testing (HbA1c, fasting glucose) often fails to detect hyperinsulinemia until late in its progression. Diagnosis requires:
Fasting serum insulin measurement
HOMA-IR calculation (fasting insulin × fasting glucose ÷ 405)
In some cases, an oral glucose tolerance test with insulin levels drawn at multiple time points
Bkaily et al. (2025) highlight that cardiovascular damage can be occurring during prediabetes — which means that waiting for glucose abnormalities to prompt insulin testing may be waiting too long.
❓ FAQ 4: Does hyperinsulinemia directly cause cancer, or is it just associated with it?
The relationship is mechanistic, not merely associative. Zhang et al. (2021) document multiple direct biological pathways through which excess insulin promotes tumor growth — including IGF-1 receptor activation, mTOR stimulation, suppression of SHBG, and chronic inflammation. While hyperinsulinemia alone rarely "causes" cancer in a deterministic sense, it creates a biological environment that significantly increases cancer risk, accelerates tumor growth, and worsens cancer prognosis. Epidemiological data from Wiebe et al. (2026) support elevated cancer-related NCD incidence in populations with co-occurring hyperinsulinemia and inflammation.
❓ FAQ 5: What dietary pattern is best for lowering insulin?
While individual responses vary, the most evidence-supported dietary approaches for reducing fasting insulin include:
Low-glycemic index diets (reduce insulin spike magnitude)
Mediterranean dietary pattern (anti-inflammatory and insulin-sensitising)
Low-carbohydrate and ketogenic diets (most effective for rapid insulin reduction in some individuals)
High-fibre, plant-forward diets (improve gut health and insulin sensitivity)
No single diet is universally superior. The key principle is reducing the frequency, magnitude, and duration of insulin surges over the course of the day.
❓ FAQ 6: Is hyperinsulinemia reversible?
Yes — particularly in its earlier stages. Since hyperinsulinemia is fundamentally a metabolic, hormonal, and lifestyle-related condition, it responds robustly to lifestyle interventions. Studies consistently show that:
Sustained weight loss of even 5–10% of body weight significantly improves insulin sensitivity
Regular physical activity can normalize fasting insulin within weeks
Dietary modifications reduce postprandial insulin spikes within days
The key message from Guess et al. (2026) is that acting early — before hyperinsulinemia has progressed to frank insulin resistance or diabetes — offers the greatest window for full metabolic restoration.
❓ FAQ 7: Should children be screened for hyperinsulinemia?
This is an evolving area. Childhood obesity rates are at historic highs in many countries, and hyperinsulinemia is increasingly recognized in pediatric populations. Children with obesity, acanthosis nigricans (dark skin patches, a sign of insulin resistance), family history of T2D, or polycystic ovary syndrome (PCOS) in adolescent girls should be discussed with a pediatrician regarding metabolic screening. Early identification and intervention in young people can prevent decades of metabolic disease and dramatically improve lifetime health outcomes.
Call to Action (CTA)
Take Control of Your Metabolic Health — Starting Today
If you've read this far, you already understand something many people don't: insulin, not just glucose, is a critical biomarker of your long-term health.
Here's how to move from awareness to action:
✅ Step 1 — Know Your Numbers Book a fasting metabolic panel with your doctor that includes fasting insulin, not just fasting glucose. Ask specifically for HOMA-IR to be calculated. Knowledge is the first act of prevention.
✅ Step 2 — Audit Your Plate For one week, pay attention to how often you consume high-glycemic foods (refined grains, sugary beverages, ultra-processed snacks). You don't need perfection — awareness is the first step to better choices.
✅ Step 3 — Move More, Strategically Add a 10-minute walk after meals. This single habit reduces postprandial glucose and insulin more effectively than the same 10 minutes of exercise at any other time of day.
✅ Step 4 — Share This Article If this information surprised you, it will likely surprise someone you care about too. Share this post with a friend, family member, or colleague who has struggled with weight, energy, or blood sugar. Collective awareness creates community-level change.
A Clinician’s Perspective: Why Hyperinsulinemia Deserves Earlier Attention
From a clinical standpoint, hyperinsulinemia represents one of the most underrecognized yet biologically significant abnormalities in modern metabolic medicine. In everyday practice, many patients present with fatigue, central obesity, hypertension, fatty liver disease, elevated triglycerides, polycystic ovary syndrome (PCOS), or prediabetes years before fasting glucose or HbA1c cross diagnostic thresholds. Increasingly, evidence suggests that chronically elevated insulin may be driving much of this pathology long before overt diabetes develops (Guess et al., 2026).
The traditional glucose-centric approach risks missing an important early intervention window. By the time hyperglycemia becomes apparent, substantial vascular, inflammatory, and metabolic dysfunction may already exist (Bkaily et al., 2025). Measuring fasting insulin, HOMA-IR, waist circumference, triglyceride-to-HDL ratio, and other markers of metabolic dysfunction can therefore provide clinically valuable insight into cardiometabolic risk at an earlier stage.
Importantly, hyperinsulinemia should not be viewed solely through the lens of diabetes prevention. Its effects extend across cardiovascular disease, non-alcoholic fatty liver disease, chronic inflammation, reproductive endocrinology, and potentially cancer biology (Zhang et al., 2021). For clinicians, this reinforces the importance of comprehensive lifestyle interventions — including nutrition, exercise, sleep optimization, and weight management — alongside evidence-based pharmacologic strategies when appropriate.
The emerging evidence argues for a shift from reactive diabetes treatment toward proactive metabolic risk detection and prevention.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Managing Diabesity: A Complete Guide to Weight Loss and Blood Sugar Control | DR T S DIDWAL
The BMI Paradox: Why "Normal Weight" People Still Get High Blood Pressure | DR T S DIDWAL
References
Bkaily, G., Jazzar, A., Abou-Aichi, A., & Jacques, D. (2025). Pathophysiology of prediabetes hyperinsulinemia and insulin resistance in the cardiovascular system. Biomedicines, 13(8), 1842. https://doi.org/10.3390/biomedicines13081842
Chawla, R. (2023). Hyperinsulinemia, obesity, and T2 diabetes: A continuum. International Journal of Diabetes in Developing Countries, 43, 171–172. https://doi.org/10.1007/s13410-023-01193-5
Guess, N., Johnson, J. D., Vaag, A., et al. (2026). Hyperinsulinaemia as a cause of obesity and cardiometabolic diseases. Nature Reviews Endocrinology. https://doi.org/10.1038/s41574-026-01240-1
Wiebe, N., Thompson, S., Stenvinkel, P., Bello, A., James, M. T., & Tonelli, M. (2026). Associations of obesity, systemic inflammation, and hyperinsulinemia with the incidence of non-communicable chronic disease and mortality: A prospective cohort study. Diabetes, Obesity and Metabolism, 28(5), 3883–3894. https://doi.org/10.1111/dom.70568
Zhang, A. M. Y., Wellberg, E. A., Kopp, J. L., & Johnson, J. D. (2021). Hyperinsulinemia in obesity, inflammation, and cancer. Diabetes & Metabolism Journal, 45(3), 285–311. https://doi.org/10.4093/dmj.2020.0250