Can You Slow Aging at the Cellular Level? The Autophagy Breakthrough Explained
New research reveals how autophagy may extend healthspan, improve metabolism, and protect against age-related diseases.
AGING
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/24/202618 min read
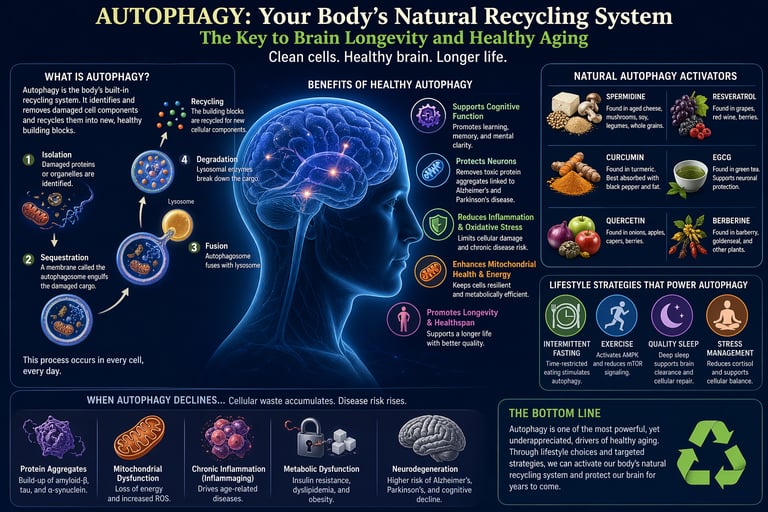
Autophagy is a natural cellular process that removes damaged proteins, dysfunctional mitochondria, and metabolic waste, recycling them into new cellular components. This process is essential for maintaining cellular health, especially in the brain, where long-lived neurons rely on efficient waste clearance. Research shows that well-functioning autophagy supports cognitive function, reduces inflammation, and lowers the risk of age-related diseases, including Alzheimer's disease.
Autophagy is regulated by key metabolic pathways, including mTOR signaling (which inhibits autophagy) and AMPK (which activates it). Lifestyle strategies such as intermittent fasting, regular exercise, quality sleep, and diets rich in polyphenols can naturally enhance autophagy.
In simple terms, autophagy acts as your body’s built-in repair system—helping slow aging, protect brain function, and extend healthspan by keeping cells clean and efficient.
Clinician’s Perspective: Autophagy & Brain–Metabolic Health
Autophagy as a central therapeutic axis:
Autophagy is increasingly recognized not merely as a cellular housekeeping process but as a modifiable determinant of aging biology. From a clinical standpoint, impaired autophagy contributes to proteotoxic stress, mitochondrial dysfunction, and systemic inflammation—core drivers of cardiometabolic and neurodegenerative disease (Bektas et al., 2025).Link to chronic disease burden:
Declining autophagic flux is mechanistically linked to conditions such as Type 2 Diabetes, Alzheimer's disease, and atherosclerotic cardiovascular disease. Clinically, this manifests as insulin resistance, sarcopenia, and cognitive decline—often coexisting in aging patients (Brand et al., 2025).Molecular targets with clinical relevance:
Key regulatory pathways—mTOR signaling and AMPK—offer actionable targets. Interventions that suppress mTOR (fasting, caloric restriction) or activate AMPK (exercise, metformin) can enhance autophagic activity and improve metabolic outcomes (Ebata & Hansen, 2026).Lifestyle prescriptions as first-line therapy:
Evidence strongly supports time-restricted eating, structured exercise, and sleep optimization as foundational strategies to preserve autophagy. These interventions are low-cost, scalable, and align with preventive medicine frameworks.Nutraceutical integration—use with caution:
Compounds such as spermidine, resveratrol, and berberine show promise in modulating autophagy pathways. However, clinicians must consider drug–nutrient interactions, dose variability, and limited long-term human data before recommending supplementation (Mundo Rivera et al., 2024).Need for biomarker-guided practice:
Routine clinical assessment of autophagy remains limited. Future practice may incorporate biomarkers such as LC3-II, p62, and lysosomal activity indices to personalize longevity interventions.Translational takeaway:
Autophagy represents a unifying mechanism bridging metabolism, neurodegeneration, and ageing. Integrating autophagy-focused strategies into clinical care may shift practice from disease treatment toward proactive healthspan optimization.
How Your Body's Recycling System Holds the Key to a Longer, Healthier Life
Your body is not just aging—it is constantly renewing itself through a highly sophisticated cellular process known as autophagy. Often described as the body’s internal “recycling and repair system,” autophagy identifies damaged proteins, dysfunctional mitochondria, and cellular debris, breaks them down, and repurposes their components to sustain cellular health. Nowhere is this process more critical than in the brain, where neurons must survive and function for decades without replacement. Emerging research suggests that the efficiency of this system may be one of the most decisive factors determining whether we age with cognitive resilience or experience decline (Bektas et al., 2025; Ebata & Hansen, 2026).
What makes this especially compelling is that autophagy is not a fixed trait—it is dynamically regulated by lifestyle, metabolism, and environmental signals. Key cellular pathways such as mTOR signaling and AMPK act as molecular switches, turning autophagy “on” or “off” depending on nutrient availability, energy status, and stress. When these pathways are optimally balanced, autophagic flux remains active, preventing the accumulation of toxic protein aggregates that are strongly linked to neurodegenerative conditions such as Alzheimer's disease and Parkinson's disease (Mundo Rivera et al., 2024).
However, with advancing age, this system becomes progressively less efficient. The decline in autophagic activity contributes to chronic inflammation, mitochondrial dysfunction, and impaired proteostasis—hallmarks of biological aging (Brand et al., 2025). The critical insight from recent science is this: aging is not merely a passive process of wear and tear, but in part a failure of cellular maintenance systems. Understanding—and strategically supporting—autophagy may therefore represent one of the most powerful, evidence-based approaches to extending not just lifespan, but healthspan.
Section 1: What Is Autophagy — and Why Should You Care?
The word autophagy comes from the Greek words autos (self) and phagein (to eat). Loosely translated, it means "self-eating" — and while that sounds alarming, it is one of the most beneficial processes occurring inside your cells every moment of every day.
Autophagy is the cellular equivalent of a recycling and waste-disposal program. When proteins misfold, when mitochondria become damaged, or when pathogens invade a cell, autophagy kicks in to engulf and break down these problematic components. The salvaged building blocks — amino acids, fatty acids, nucleotides — are then reused to build new, healthy cellular structures.
According to Vijay and Varshney (2025) in the Springer volume Rejuvenating the Brain: Nutraceuticals, Autophagy, and Longevity, this process is not a passive bystander in brain health — it is a central, active architect of neural integrity. The brain, with its extraordinarily high metabolic demand and post-mitotic (non-dividing) neurons, depends on autophagy more critically than almost any other tissue in the body. Neurons that cannot clear their own cellular debris accumulate toxic aggregates — the very hallmark of Alzheimer's, Parkinson's, and other devastating neurodegenerative conditions (Vijay & Varshney, 2025).
The Three Main Forms of Autophagy
Not all autophagy is the same. Researchers distinguish three primary types:
Macroautophagy: The most well-studied form. A double-membrane structure called the autophagosome engulfs cargo and fuses with the lysosome for degradation. When people refer to "autophagy," they typically mean this form.
Microautophagy: The lysosome directly engulfs small cytoplasmic components through membrane invagination.
Chaperone-Mediated Autophagy (CMA): A highly selective process where specific proteins bearing a recognition motif are unfolded and transported directly across the lysosomal membrane by chaperone proteins. CMA is particularly critical for aging research, as it selectively targets damaged or misfolded proteins linked to neurodegeneration.
Section 2: Autophagy and the Aging Brain — The Critical Connection
Here is the sobering truth: autophagy naturally declines with age. The cellular machinery that powers this recycling system — autophagy-related proteins (ATGs), lysosomal enzymes, and regulatory pathways like mTOR and AMPK — all show measurable functional decline as decades pass. The consequences for brain health are profound.
A landmark 2025 study published in Aging Cell by Bektas et al. (2025) examined autophagy across a broad population cohort and found a striking pattern: individuals who maintained robust autophagic function as they aged showed significantly better metabolic and cellular health profiles consistent with what the researchers term "healthy aging." Critically, the study demonstrated that the preservation of autophagy — not just its presence, but its active maintenance over time — may be a defining biological mechanism separating healthy agers from those who experience accelerated decline.
This is more than an interesting correlation. Bektas and colleagues (2025) found that autophagic preservation was associated with markers of reduced systemic inflammation, better mitochondrial function, and healthier protein homeostasis (proteostasis) — all critical pillars of brain longevity.
Complementing this, a comprehensive 2026 review by Ebata and Hansen, published in the Journal of Molecular Biology, synthesized mounting evidence linking autophagy to virtually every dimension of healthy aging at the molecular level. Their analysis highlights how autophagic flux — the dynamic, ongoing throughput of the autophagy pathway — must remain active and responsive to maintain neuronal health. When flux stalls, misfolded proteins and dysfunctional organelles accumulate, triggering inflammation, oxidative stress, and ultimately cell death (Ebata & Hansen, 2026).
Key Research Insight
Individuals who maintain higher autophagic activity as they age demonstrate measurably better cognitive resilience, reduced neuroinflammation, and lower rates of proteotoxic accumulation — the build-up of damaged proteins that drives Alzheimer's and Parkinson's disease pathology (Bektas et al., 2025; Ebata & Hansen, 2026).
Section 3: When Autophagy Fails — The Metabolic and Neurological Cost
Understanding what happens when autophagy falters is just as important as understanding what it does when it works well.
Brand, Ntuli, and Loos (2025), writing in Expert Opinion on Therapeutic Targets, provide a rigorous analysis of "autophagic failure with age" — documenting how declining autophagy is not merely a passive consequence of aging, but an active contributor to metabolic disorders. Their review demonstrates that autophagic insufficiency accelerates insulin resistance, promotes lipid accumulation, drives mitochondrial dysfunction, and fuels chronic low-grade inflammation (inflammaging) — all of which compound neurodegeneration and reduce cognitive longevity.
The clinical implications are stark. Conditions now understood to involve compromised autophagy include:
Alzheimer's Disease: Accumulation of amyloid-beta plaques and tau tangles — hallmarks of Alzheimer's — is substantially driven by impaired autophagic clearance.
Parkinson's Disease: Alpha-synuclein aggregates that destroy dopaminergic neurons resist clearance when CMA is dysfunctional.
Type 2 Diabetes and Metabolic Syndrome: Autophagic failure in pancreatic beta cells and liver cells accelerates insulin resistance.
Cardiovascular Disease: Impaired mitophagy (autophagy of mitochondria) in cardiac cells promotes heart disease.
Brand et al. (2025) argue compellingly that therapeutic strategies targeting autophagy restoration — rather than only treating downstream symptoms — represent one of the most promising frontiers in age-related disease medicine. Their paper explicitly calls for development of autophagy-targeting interventions as first-line preventive strategies.
Section 4: Natural Autophagy Activators — What the Evidence Says
One of the most exciting and actionable frontiers in longevity research is the identification of natural compounds — many found in everyday foods — that potently activate autophagy. A landmark 2024 systematic review in the journal Cells by Mundo Rivera et al. (2024) provides the most comprehensive mapping of natural autophagy activators to date, specifically evaluated in the context of fighting age-related diseases.
Here are the most evidence-supported natural autophagy activators identified in current research:
1. Spermidine
Perhaps the most compelling natural autophagy inducer currently under investigation. Found in aged cheese, mushrooms, whole grains, legumes, and soy products, spermidine is a polyamine that activates autophagy through epigenetic mechanisms — specifically by inhibiting histone acetyltransferase EP300. Mundo Rivera et al. (2024) highlight robust evidence linking spermidine supplementation to extended lifespan in model organisms and neuroprotective effects in early human trials. Critically, spermidine's autophagy-inducing effects are mTOR-independent, making it uniquely valuable because mTOR inhibition (e.g., from rapamycin or fasting) carries unwanted side effects in long-term use.
2. Resveratrol
Found in red wine, grapes, and berries, resveratrol activates the SIRT1 deacetylase pathway, which in turn deacetylates and activates key autophagy proteins. Mundo Rivera et al. (2024) document resveratrol's capacity to promote autophagic flux specifically in neuronal cells, with demonstrated neuroprotective effects in models of Alzheimer's and Parkinson's disease. Its ability to cross the blood-brain barrier makes it particularly relevant for brain longevity.
3. Curcumin
The active compound in turmeric, curcumin is one of the most studied natural autophagy inducers. It activates autophagy through AMPK pathway stimulation and mTOR inhibition, while simultaneously suppressing the neuroinflammatory NF-κB pathway. The dual action — pro-autophagy and anti-inflammatory — makes curcumin exceptionally relevant to brain aging. Mundo Rivera et al. (2024) note that bioavailability remains the primary clinical challenge, with piperine (from black pepper) and lipid-based formulations substantially improving absorption.
4. Epigallocatechin Gallate (EGCG)
The primary bioactive catechin in green tea, EGCG activates autophagy through AMPK signaling. It has demonstrated protective effects against amyloid-beta toxicity in neuronal models and shows anti-aggregation properties against both amyloid-beta and tau proteins — the two primary pathological drivers of Alzheimer's disease. Regular green tea consumption is associated epidemiologically with reduced dementia risk in multiple large cohort studies.
5. Quercetin
A flavonoid found abundantly in onions, apples, capers, and berries, quercetin activates autophagy while simultaneously exerting senolytic effects — selectively clearing senescent ("zombie") cells that accumulate with age and drive chronic inflammation. Mundo Rivera et al. (2024) highlight quercetin as one of the few compounds with demonstrated activity across both autophagy induction and senescent cell clearance, representing a dual anti-aging mechanism.
6. Berberine
An alkaloid found in plants like barberry and goldenseal, berberine is a potent AMPK activator. Its autophagy-inducing effects are well-documented, and emerging research specifically links berberine to neuroprotection. Particularly notable is berberine's demonstrated ability to reduce amyloid-beta production and tau phosphorylation in preclinical models, suggesting direct relevance to Alzheimer's prevention.
Patient Safety Note
While these natural compounds are generally considered safe at dietary levels, supplementation doses may interact with medications. Always consult your healthcare provider before starting any supplement regimen, particularly if you take blood thinners, diabetes medications, or immunosuppressants. This article is for educational purposes and does not constitute medical advice.
Section 5: Lifestyle Strategies That Powerfully Preserve Autophagy
Natural compounds are powerful, but lifestyle interventions may be even more foundational. Here are the evidence-based lifestyle strategies most strongly linked to autophagy preservation and brain longevity:
Intermittent Fasting and Caloric Restriction
Fasting is the most potent known inducer of autophagy. When nutrients are scarce, mTORC1 activity drops and AMPK activity rises — a metabolic shift that powerfully upregulates autophagic flux. Even 16–24 hours of fasting shows measurable autophagy induction in human studies. Time-restricted eating (e.g., 16:8 intermittent fasting) has emerged as a practical, sustainable approach that activates autophagy without severe caloric restriction. Vijay and Varshney (2025) emphasize fasting as foundational to any brain longevity protocol, given the brain's particular vulnerability to proteotoxic accumulation.
Exercise — Particularly Aerobic and Resistance Training
Physical exercise — especially endurance exercise — is a potent autophagy inducer through both AMPK activation and reduced mTOR signaling. Bektas et al. (2025) note that physically active individuals in their dataset showed more preserved autophagic signatures, consistent with a large body of evidence linking exercise to brain longevity. Resistance training adds the additional benefit of improving insulin sensitivity, reducing a key driver of autophagic inhibition (chronic hyperinsulinemia suppresses autophagy via sustained mTOR activation).
Sleep Quality and Circadian Rhythm Alignment
Autophagy in the brain follows circadian rhythms, with peak activity occurring during sleep — particularly during the deep sleep phases when the glymphatic system (the brain's waste clearance system) is most active. Chronic sleep deprivation or circadian disruption significantly impairs autophagic activity. Ebata and Hansen (2026) specifically discuss the circadian regulation of autophagy as a critical and under-recognized mechanism in brain aging.
Dietary Patterns — The Mediterranean and MIND Diets
Diets rich in polyphenols, healthy fats (olive oil, nuts), and low in ultra-processed foods support autophagic activity through multiple mechanisms — providing natural autophagy-activating compounds (resveratrol, quercetin, EGCG), reducing metabolic stress, and supporting gut microbiome diversity. The Mediterranean diet, in particular, provides high dietary spermidine from legumes, whole grains, and fermented foods.
Stress Reduction and Hormetic Stress
Paradoxically, mild, controlled stressors — a concept called hormesis — robustly activate autophagy. Cold exposure, heat stress (sauna), and moderate-intensity exercise all qualify as hormetic stressors. Conversely, chronic psychological stress, through sustained cortisol elevation, impairs autophagic function. Mind-body practices — yoga, meditation, breathwork — that reduce chronic stress, therefore indirectly support autophagic health.
Section 6: Practical Applications — Your Autophagy Action Plan
Translating science into daily life is where real health transformation happens. Based on the cumulative evidence reviewed above, here is a practical, tiered action plan:
Tier 1: Start This Week (Foundation)
1. Eat within a 10–12 hour window daily. Gradually narrow to 8 hours if tolerated. This is the simplest, most evidence-backed autophagy activator available.
2. Add green tea (2–4 cups/day). Choose high-quality matcha or sencha for maximum EGCG content.
3. Include turmeric in cooking daily — add with black pepper and a fat source to maximize curcumin absorption.
4. Move for 30–45 minutes daily. Even brisk walking triggers autophagic signaling. Aim for 3 resistance sessions per week.
5. Prioritize 7–9 hours of sleep. Keep consistent sleep and wake times to support circadian autophagy rhythms.
Tier 2: Optimize Over 30 Days
6. Shift toward a Mediterranean-style diet. Increase legumes (spermidine), colourful vegetables (quercetin, resveratrol precursors), and extra-virgin olive oil (oleocanthal).
7. Consider 24-hour fasts once per month if cleared by your physician. This provides a powerful autophagic reset.
8. Incorporate heat or cold hormesis: 2–3 sauna sessions per week OR cold showers (30–90 seconds cold at end of shower).
9. Reduce ultra-processed foods, refined sugars, and alcohol — all of which impair autophagic function and accelerate cellular aging.
Tier 3: Discuss With Your Healthcare Provider
10. Targeted nutraceutical supplementation (spermidine, berberine, resveratrol, quercetin with bromelain) based on individual health status and medication interactions.
11. Comprehensive metabolic and inflammatory biomarker testing to assess your current autophagic and aging phenotype.
12. Pharmacological options (e.g., metformin, rapamycin) being actively investigated for their autophagy-preserving and longevity-promoting effects — evidence is accumulating but protocols are still emerging (Brand et al., 2025).
Frequently Asked Questions About Autophagy and Brain Longevity
❓ Q1. What is autophagy in simple terms?
Think of autophagy as your cells' built-in housekeeping service. It identifies damaged or no-longer-needed cellular components — broken proteins, worn-out mitochondria, invading pathogens — packages them up, and recycles them into usable building materials. This keeps your cells clean, efficient, and youthful. In your brain specifically, this process is critical because brain neurons must survive for an entire lifetime and cannot simply be replaced the way skin cells are.
❓ Q2. Does autophagy really slow brain aging?
The scientific consensus is building strongly in favor of this conclusion. Bektas et al. (2025), publishing in Aging Cell, found that preserved autophagic function is a distinguishing feature of healthy aging. Ebata and Hansen (2026) synthesized extensive molecular evidence linking autophagic flux to neuronal health and cognitive preservation. Vijay and Varshney (2025) specifically frame autophagy as a central mechanism of brain longevity. While human longevity research is inherently complex, the mechanistic and epidemiological evidence is compelling.
❓ Q3. Can I really activate autophagy with food alone?
Yes — significantly so. Mundo Rivera et al. (2024) conducted a comprehensive review of natural autophagy activators and found robust evidence for compounds like spermidine (in aged cheese, legumes, mushrooms), resveratrol (red grapes, berries), curcumin (turmeric), EGCG (green tea), quercetin (onions, apples), and berberine (barberry root). These compounds activate autophagy through well-characterized molecular pathways. Dietary patterns rich in these compounds — such as the Mediterranean diet — are consistently associated with better cognitive aging outcomes.
❓ Q4. Does fasting really trigger autophagy? How long do I need to fast?
Yes, fasting is the most powerful known trigger of autophagy. Nutrient deprivation drops mTORC1 activity (the primary autophagy inhibitor) and raises AMPK activity (the primary autophagy activator). Meaningful autophagic upregulation begins to occur within 12–16 hours of fasting, with more robust effects seen with 24-hour fasts. However, even time-restricted eating (eating within an 8–10 hour window daily) shows measurable autophagic benefits and is sustainable long-term without the caloric restriction risks associated with prolonged fasting.
❓ Q5. What happens when autophagy fails as we age?
Brand, Ntuli, and Loos (2025) in Expert Opinion on Therapeutic Targets document the downstream consequences clearly: autophagic failure accelerates metabolic dysfunction (insulin resistance, dyslipidemia), drives mitochondrial decline, promotes chronic systemic inflammation (inflammaging), and allows proteotoxic aggregates to accumulate in neurons. These downstream effects collectively accelerate cognitive decline, increase Alzheimer's and Parkinson's risk, and worsen cardiovascular and metabolic health. Crucially, autophagic failure is now understood to be not merely a side effect of aging, but an active driver of it.
❓ Q6. Are autophagy supplements safe? Should I take them?
Many natural autophagy activators are safe at dietary doses and have excellent long-term safety profiles. Spermidine, resveratrol, quercetin, berberine, and curcumin have all been studied in human trials with favorable safety data. However, supplements can interact with medications — berberine and metformin both lower blood sugar and may have additive effects; resveratrol interacts with blood thinners; quercetin may affect thyroid hormone absorption. Always discuss supplementation with your primary care provider, especially if you manage chronic conditions or take prescription medications. Start with dietary sources wherever possible.
❓ Q7. Is there a single most important thing I can do to support autophagy and brain health?
If forced to choose one intervention with the broadest, most consistent evidence base, it would be the combination of time-restricted eating (16:8 fasting protocol) with regular aerobic exercise. Together, these two lifestyle modifications create powerful AMPK activation and mTOR suppression — the core biochemical lever of autophagy induction. They are free, accessible, medication-free, and supported by decades of human research. Adding dietary polyphenols (green tea, turmeric, berries) and optimizing sleep quality amplifies these benefits substantially.
The Autophagy Blueprint: Your Daily Protocol for Brain Longevity
This protocol is designed to synchronize your lifestyle with your body's natural cellular recycling rhythms. By strategically timing nutrients and stressors, you maximize autophagic flux—the process that clears neurotoxic proteins before they can accumulate.
Morning: The "Recycle & Hydrate" Phase
The Ritual: Start with a cup of high-quality Green Tea (Matcha or Sencha).
The Science: The EGCG in green tea stimulates the AMPK pathway, prolonging the autophagic window initiated by your overnight fast.
The Action: Complete a 16-hour fast. When you do break it, prioritize "mindful re-feeding"—avoiding high-sugar "glucose spikes" that immediately shut down the repair process.
Mid-Day: The "Synaptic Shield" Meals
The Ritual: Incorporate the "Longevity Trio" (Turmeric + Black Pepper + Healthy Fat) into at least one meal.
The Science: Curcumin is a potent neuro-protective agent, but it requires piperine (pepper) and lipids for absorption to reach the blood-brain barrier.
The Action: Use turmeric in soups, stews, or dressings. Aim for Spermidine-rich additions like mushrooms, lentils, or fermented soy to keep the "cleaning crew" active even after eating.
Afternoon: The "Hormetic Pulse"
The Ritual: Commit to a 30-minute brisk walk or resistance training session.
The Science: Exercise is a "hormetic stressor"—a beneficial stress that shocks cells into a defensive, self-cleaning mode while improving insulin sensitivity.
The Action: If you spend your day at a desk, think of this as your "metabolic reset." It clears out cellular debris generated by the day's mental load.
Evening: The "Glymphatic Wash"
The Ritual: Establish a strict Digital Sunset and consistent sleep window.
The Science: Your brain doesn't just rest; it physically "washes" itself. The glymphatic system flushes out metabolic waste (like amyloid-beta) specifically during deep, NREM sleep.
The Action: Stop eating at least 3 hours before bed. This ensures insulin levels are low, allowing the "Night Shift" cleaning crew to work at maximum capacity.
Pro-Tip: Autophagy is not a "once-and-done" event; it is a biological habit. The more consistent your rhythm, the more efficient your cellular housekeeping becomes. Start with one "Green" habit today and layer the rest over the next 30 days.
Clinical pearls
1. The "Lysosomal Bottleneck" Effect
Scientific Tone: Autophagic health is not just about induction (starting the process) but about flux (completing it). As we age, the lysosome’s pH can rise, making its enzymes less acidic and less effective at breaking down waste. If the "trash" is bagged but not burned, it creates cellular congestion.
Patient-Friendly Tone: Think of your cells like a house. It’s not enough to just put the trash in bags; the garbage truck actually has to take it away. To keep your "trucks" running, you need deep sleep and a healthy metabolism so the trash doesn't just pile up in the driveway.
2. Spermidine as a "Muscle-Safe" Activator
Scientific Tone: Unlike rapamycin or extreme caloric restriction, which inhibit mTOR (the pathway for muscle growth), spermidine induces autophagy via EP300 inhibition. This allows for the activation of cellular recycling without necessarily suppressing the anabolic signals needed to maintain muscle mass.
Patient-Friendly Tone: Most "cleanses" can make you lose muscle along with the junk. Spermidine (found in mushrooms and legumes) is a "smart" cleaner. It tells the cell to recycle its internal trash but doesn't flip the "off" switch on your muscles, making it a great tool for staying strong while staying clean.
3. The "Metabolic Switch" Threshold
Scientific Tone: Autophagy is governed by the AMPK/mTOR rheostat. Meaningful upregulation typically requires a transition from glucose-burning to fatty acid oxidation. Chronic hyperinsulinemia keeps mTOR perpetually active, effectively "locking" the door to the autophagy pathway.
Patient-Friendly Tone: Your body has a "cleaning switch." When your insulin is high (from constant snacking or sugar), that switch is stuck in the "off" position. By giving your body breaks from eating, you flip the switch to "on," allowing your cells to finally start their internal repairs.
4. Circadian-Glymphatic Synchronization
Scientific Tone: The brain’s glymphatic system (the physical wash) and autophagy (the internal recycling) are both under circadian control. Peak autophagic flux in the brain occurs during deep NREM sleep. Disruption of these rhythms causes "proteotoxic stress," where damaged proteins like amyloid-beta accumulate simply because the cleaning crew was "called off" their shift.
Patient-Friendly Tone: Your brain has a "night shift." While you sleep, your cells do their deepest cleaning. If you don't sleep well or you stay up late under bright lights, you're essentially firing your cleaning crew. Consistent sleep is the best "brain wash" money can't buy.
5. Hormetic Stress: The "Use It or Lose It" Signal
Scientific Tone: Autophagy is a survival mechanism. Hormesis—brief, controlled exposure to stressors like heat (sauna), cold, or high-intensity exercise—triggers the SIRT1 pathway. This "shocks" the cell into a defensive mode that prioritizes structural repair and mitochondrial renewal.
Patient-Friendly Tone: Our bodies were designed for a world that was sometimes too hot, too cold, or physically demanding. Modern comfort makes our cells "lazy." By taking a cold shower or using a sauna, you’re giving your cells a "fire drill" that keeps the recycling system sharp and ready for action.
6. The Bioavailability "Golden Rule" for Curcumin
Scientific Tone: Curcumin is a potent autophagy inducer, but its lipophilic nature and rapid metabolism lead to poor systemic bioavailability. For clinical efficacy in neuroprotection, it must be administered with a glucuronidation inhibitor (like piperine) or in a lipid-micellar formulation to ensure it reaches the blood-brain barrier.
Patient-Friendly Tone: Turmeric is a superstar for your brain, but your body is very bad at absorbing it. If you just eat the powder, it mostly goes right through you. Always mix turmeric with a little black pepper and a healthy fat (like olive oil or avocado) to make sure the "medicine" actually gets into your bloodstream and up to your brain.
Author’s Note
This article was written with a clear purpose: to bridge the gap between rapidly evolving molecular science and real-world clinical application. The field of autophagy has expanded dramatically over the past decade, and particularly in the 2024–2026 period, with growing recognition of its central role in aging, metabolism, and brain health. Yet, much of this knowledge remains confined to specialized journals, often inaccessible to both patients and busy clinicians.
As a physician, I have increasingly observed that many chronic conditions we treat—whether Type 2 Diabetes, Alzheimer's disease, or cardiovascular disease—share common underlying mechanisms rooted in impaired cellular maintenance and metabolic dysregulation. Autophagy represents one of the most compelling unifying pathways linking these conditions. Understanding it not only deepens our insight into disease pathophysiology but also opens the door to more upstream, preventive strategies.
However, it is equally important to acknowledge the current limitations of the evidence. While mechanistic and preclinical data are robust, and human observational studies are promising, many interventions discussed—particularly nutraceuticals and pharmacological modulators—are still under active investigation. Clinical decisions must therefore remain individualized, evidence-informed, and grounded in patient safety.
This work aims to translate complex biology into practical, actionable knowledge without oversimplifying the science. Readers are encouraged to view autophagy not as a single intervention target, but as part of a broader systems-based approach to health—integrating nutrition, physical activity, sleep, and metabolic care.
Finally, science in this domain is evolving rapidly. What we understand today will continue to refine in the coming years. Staying curious, critical, and evidence-driven is essential—for clinicians and patients alike.
This article is intended for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before beginning a new exercise or nutrition program, especially if you have an existing medical condition.
Related Articles
Why Do Muscles Weaken With Age? The Mitochondria Link Explained
Can You Slow Brain Aging? Microglia and Neuroinflammation Explained
Neuromuscular Aging Explained: How Nerve–Muscle Breakdown Causes Weakness After 40
The mTOR Switch: Why Your Cells Stopped Listening to Insulin | DR T S DIDWAL
Can Resistance Training Reverse Diabetes? The Muscle–mTOR Link | DR T S DIDWAL
References
Bektas, A., Schurman, S. H., Candia, J., Santiago-Fernández, O., Kaushik, S., Cuervo, A. M., & Ferrucci, L. (2025). Preservation of autophagy may be a mechanism behind healthy aging. Aging Cell, 24, e70246. https://doi.org/10.1111/acel.70246
Brand, E., Ntuli, M., & Loos, B. (2025). Autophagic failure with age: Influence on metabolic disorders and prospects for therapeutic targeting. Expert Opinion on Therapeutic Targets, 29(10), 677–686. https://doi.org/10.1080/14728222.2025.2584012
Ebata, H., & Hansen, M. (2026). Links between autophagy and healthy aging. Journal of Molecular Biology, 438(6), 169656. https://doi.org/10.1016/j.jmb.2026.169656
Mundo Rivera, V. M., Tlacuahuac Juárez, J. R., Murillo Melo, N. M., Leyva Garcia, N., Magaña, J. J., Cordero Martínez, J., & Jiménez Gutierrez, G. E. (2024). Natural autophagy activators to fight age-related diseases. Cells, 13(19), 1611. https://doi.org/10.3390/cells13191611
Vijay, A., & Varshney, R. (2025). Autophagy and brain longevity. In A. Kumar Singh & S. N. Rai (Eds.), Rejuvenating the brain: Nutraceuticals, autophagy, and longevity (Nutritional Neurosciences series, pp. Ch. 3). Springer. https://doi.org/10.1007/978-981-95-2790-8_3