What Happens Inside Your Muscles During a Workout
Exercise does more than burn calories. Learn how workouts reprogram metabolism, trigger muscle growth, and strengthen immunity at the cellular level.
EXERCISE
Dr. T.S. Didwal, M.D.(Internal Medicine)
2/25/202616 min read
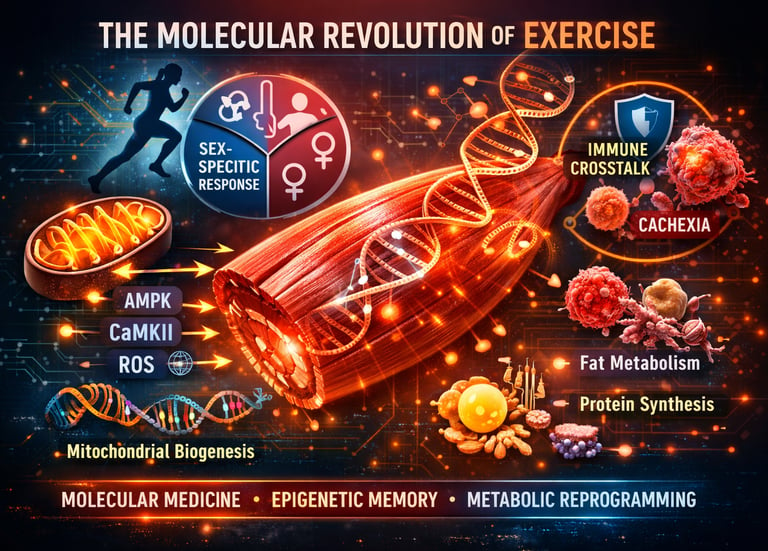
What Your Muscles Are Really Doing During a Workout (It’s Not Just Burning Calories)” It is about rewriting your cellular code?
For decades, exercise science focused on macroscopic outcomes: VO₂ max, muscle size, fat loss, and strength gains. But modern multi-omics research, skeletal muscle transcriptomics, and mitochondrial proteomics now reveal something far more profound. Exercise is a form of molecular medicine — a stimulus that activates hierarchical metabolic signaling pathways, reprograms epigenetic architecture, and reshapes systemic immune–metabolic crosstalk (Smith et al., 2023).
Inside contracting muscle fibers, energetic stress activates AMP-activated protein kinase (AMPK), calcium flux stimulates CaMKII, and controlled bursts of reactive oxygen species (ROS) function as adaptive signaling molecules rather than damage agents. These signals converge on transcriptional regulators like PGC-1α, driving mitochondrial biogenesis, oxidative phosphorylation efficiency, and long-term metabolic remodeling (Weidenhamer et al., 2025).
But adaptation is not uniform. Large-scale sex-stratified multi-omics integration demonstrates that males and females activate distinct molecular programs in response to identical exercise stimuli, influencing lipid metabolism, extracellular matrix remodeling, and anabolic signaling networks (Jacques et al., 2025). Even more striking, exercise shares metabolic nodes with disease states such as cachexia, differing not in pathway activation but in the critical resolution of inflammatory and catabolic stress (Westermann et al., 2026).
This is the new paradigm: skeletal muscle is not merely a contractile tissue. It is an endocrine organ, an epigenetic memory bank, and a systemic metabolic regulator. Every workout is a precisely dosed biological signal — capable of reprogramming metabolism, immunity, and resilience at the cellular level.
Clinical pearls
1. Exercise is a "Molecular Message," Not Just a Calorie Burner
Scientific Perspective: Contraction triggers the release of myokines (like IL-6 and Irisin) and activates the AMPK pathway. This shifts the body from a pro-inflammatory state to an immunomodulatory, anti-inflammatory state by communicating directly with the liver, brain, and adipose tissue.
"Think of exercise as a natural pharmacy. Every time you move, your muscles 'mail' signals to the rest of your body to lower inflammation and improve your mood, regardless of how many calories you see on your smartwatch."
2. The "Metabolic Savings Account" (Muscle Memory)
Scientific Perspective: As shown by Weidenhamer et al. (2025), prior training creates persistent epigenetic marks (DNA methylation) on mitochondrial genes. This "primes" the muscle, allowing for a faster VO2 max recovery and hypertrophic response during retraining.
"No workout is ever wasted. Even if you’ve taken a long break due to injury or life, your muscles have a 'savings account' of past hard work. When you start again, your body will recognize the stimulus and bounce back much faster than a beginner's would."
3. Sex is a Biological Variable, Not a Confounding One
Scientific Perspective: The multi-omics data from Jacques et al. (2025) reveal that females have a more robust transcriptomic response in lipid metabolism and extracellular matrix pathways, while males show higher upregulation in glycolytic and contractile protein synthesis.
"Men and women may respond differently to the same workout. A woman’s body might be more efficient at switching to fat-burning and tissue repair, while a man’s might focus more on raw power and sugar metabolism. We can tailor your plan to work with your biology, not against it."
4. The "Anabolic Window" of Resolution in Chronic Disease
Scientific Perspective: Westermann et al. (2026) highlight that while both exercise and cachexia (wasting) involve inflammation, exercise includes a resolution phase (anabolic rebound). In cachexia, this resolution is broken, leading to a "leaky" metabolic state.
"In chronic illness, the body is stuck in 'breakdown mode.' Controlled exercise acts as a reset switch. By creating a brief, healthy stress followed by rest, we are teaching your body how to finish the repair process that the disease is trying to block."
Part 1: Exercise Metabolism and Skeletal Muscle Adaptation — The Molecular Blueprint
The publication of Smith et al. (2023) in Nature Reviews Molecular Cell Biology was a watershed moment for exercise science. This comprehensive review established a definitive molecular blueprint of how skeletal muscle senses, responds to, and adapts to exercise stimuli — integrating decades of fragmented findings into a coherent, unified framework.
At its core, skeletal muscle is not merely a contractile tissue. It is a metabolically dynamic organ that continuously interprets mechanical and biochemical signals from its environment. Smith and colleagues describe how different forms of exercise — endurance versus resistance, for instance — activate distinct but overlapping molecular cascades. Endurance exercise predominantly activates pathways associated with mitochondrial biogenesis, fatty acid oxidation, and oxidative phosphorylation. Resistance exercise, by contrast, drives anabolic signalling through the mechanistic target of rapamycin complex 1 (mTORC1), stimulating protein synthesis and myofibrillar remodelling.
One of the review's most valuable contributions is its articulation of the exercise sensor hierarchy. Key among these sensors is AMP-activated protein kinase (AMPK), which detects energetic stress — the ratio of AMP/ATP — and orchestrates a cellular response that prioritises energy conservation and metabolic efficiency. Equally important is the calcium-dependent activation of calmodulin-dependent protein kinase II (CaMKII), which links excitation–contraction coupling to downstream transcriptional changes. The authors also highlight the role of reactive oxygen species (ROS), which, at physiological levels generated during exercise, serve as critical signalling molecules rather than merely damaging by-products.
Perhaps most importantly for translational medicine, Smith et al. (2023) emphasise that these molecular adaptations are highly dose-dependent, time-sensitive, and context-specific. A single bout of exercise produces a transient gene expression response; repeated bouts over weeks and months drive epigenetic reprogramming that locks in long-term structural and functional changes. This chronic remodelling encompasses not only contractile proteins but also metabolic enzymes, membrane transporters, and even the muscle's vascular architecture.
Skeletal muscle adaptation to exercise is orchestrated by a hierarchical network of molecular sensors — including AMPK, CaMKII, and ROS — that translate mechanical and metabolic signals into precise, long-lasting structural and functional changes. Endurance and resistance exercise activate distinct yet partially overlapping pathways, making exercise modality a critical variable in any training or therapeutic programme.
Part 2: Molecular Landscape of Sex- and Modality-Specific Adaptation — A Multi-Omics Revolution
The study byJacques et al. (2025 )' is about sex differences in molecular adaptation. Despite decades of exercise physiology research, sex has historically been treated as a confounding variable rather than a biological factor of primary interest. correct this oversight emphatically. They demonstrate that males and females differ not only in the magnitude of adaptation but in the qualitative nature of the molecular programmes activated. Females show more pronounced transcriptomic responses in pathways related to lipid metabolism and extracellular matrix remodelling, while males exhibit stronger upregulation of contractile protein synthesis and glycolytic enzyme expression following resistance training.
The multi-omics integration approach — simultaneously measuring changes across DNA methylation, gene expression, protein abundance, and metabolite profiles — allowed the authors to identify molecular hubs that coordinate adaptation across biological layers. This is a critical advancement, because single-omics studies often miss the emergent properties that arise from cross-layer interactions. The study identified several key transcription factors and regulatory non-coding RNAs that appear to function as master orchestrators of sex-specific adaptation, many of which had not previously been implicated in exercise biology.
Equally important are the modality-specific findings. Comparing high-intensity interval training (HIIT) with moderate-intensity continuous training (MICT) and resistance training, the authors show that each modality generates a distinct molecular signature. HIIT produced the broadest transcriptional response and the greatest degree of mitochondrial pathway upregulation, consistent with its known efficacy for cardiorespiratory fitness. Resistance training was uniquely associated with large-scale proteome remodelling, particularly of sarcomeric proteins. These findings have immediate clinical relevance: they suggest that exercise prescriptions can and should be personalised not only by fitness goal but also by biological sex.
Using large-scale multi-omics integration in human skeletal muscle, this study reveals that sex is a primary — not secondary — determinant of molecular adaptation to exercise. Males and females activate qualitatively different molecular programmes in response to the same training stimuli, and each exercise modality (HIIT, MICT, resistance) generates a distinct molecular signature. Personalised exercise prescription based on sex and modality is not merely preferable; it is molecularly justified.
Part 3: Muscle Memory of Exercise — How Past Training Shapes Future Metabolism
The concept of "muscle memory" is familiar to most people in the context of motor learning — the idea that once you learn to ride a bike, your muscles seem to remember how to do it even after years of inactivity. But Weidenhamer et al. (2025) explore a deeper, more biochemical form of muscle memory: the epigenetic encoding of metabolic adaptation that persists long after a training programme ends.
Published in the American Journal of Physiology–Cell Physiology, this study demonstrates that prior exercise training leaves a lasting imprint on mitochondrial gene expression and function — an imprint that fundamentally reshapes how muscle responds to subsequent anabolic stimuli. Using a detraining-and-retraining experimental model, the authors show that muscles with a prior exercise history mount a faster, more robust mitochondrial response upon re-exposure to training than muscles encountering exercise for the first time. Critically, this accelerated response translates into enhanced skeletal muscle growth, linking mitochondrial metabolic efficiency directly to hypertrophic capacity.
The mechanism, the study proposes, is largely epigenetic. Prior exercise induces specific DNA methylation and histone modification patterns at mitochondrial biogenesis genes — including PGC-1α and components of the electron transport chain — that remain partially in place even after detraining. When exercise is resumed, these "primed" loci are activated more rapidly and to a greater extent than in naïve muscle, creating a metabolic advantage that supports not only energy production but also the biosynthetic demands of muscle protein synthesis.
This research has profound implications for rehabilitation medicine, athletic return-to-play protocols, and the management of chronic diseases associated with muscle loss. It suggests that any prior exercise history — even in patients who have been deconditioned for months or years — confers a molecular advantage that can be leveraged upon reintroduction of training.
The study also introduces an elegant conceptual bridge between mitochondrial metabolism and muscle hypertrophy — two processes that have traditionally been studied in isolation. By showing that mitochondrial efficiency is a prerequisite for optimal anabolic signalling rather than simply a parallel outcome, Weidenhamer and colleagues challenge the conventional wisdom that endurance and hypertrophy adaptations are inherently at odds.
Prior exercise training encodes a lasting epigenetic memory at mitochondrial biogenesis loci, priming muscle for faster and more robust metabolic adaptation upon retraining. This mitochondrial "muscle memory" directly enhances the capacity for skeletal muscle growth, bridging the traditionally separate domains of endurance and hypertrophy adaptation. The findings support leveraging exercise history in rehabilitation and chronic disease management.
Part 4: Muscle–Immune Metabolic Crosstalk — The Bridge Between Exercise and Disease
Published in Current Opinion in Biotechnology in 2026, the review by Westermann and colleagues tackles one of the most clinically urgent questions in exercise science: how do the metabolic conversations between skeletal muscle and the immune system differ between pathological muscle wasting (cachexia) and the adaptive muscle remodelling driven by exercise? And, crucially, where do these two states overlap?
Cachexia — the severe muscle and fat wasting that accompanies cancer, heart failure, chronic kidney disease, and other systemic conditions — remains one of the most devastating and poorly treated clinical syndromes in medicine, accounting for approximately 20–30% of all cancer deaths. Its molecular hallmarks include sustained inflammatory cytokine signalling (particularly TNF-α, IL-6, and IL-1β), mitochondrial dysfunction, impaired protein synthesis, and accelerated autophagy. These features are, to varying degrees, also present in skeletal muscle during and immediately after intense exercise — a paradox that Westermann et al. (2026) dissect with precision.
The review identifies a set of shared metabolic nodes between cachexia and exercise, including altered amino acid metabolism (particularly branched-chain amino acid catabolism), perturbations in the NAD⁺/NADH ratio, and activation of the unfolded protein response (UPR) in the endoplasmic reticulum. The critical difference, the authors argue, lies in the resolution of these shared stresses. In exercise, the inflammatory and catabolic signals are transient and followed by an anabolic recovery phase; in cachexia, they are sustained and unresolved, preventing the tissue repair cascade from completing.
The concept of myokines — cytokines secreted by contracting muscle — features prominently in this analysis. Exercise-induced myokines such as IL-6, irisin, and BDNF have immunomodulatory properties that, when transiently elevated, promote anti-inflammatory polarisation of macrophages and regulatory T cell activity. In cachexia, chronic low-grade inflammation corrupts these signalling loops, converting what should be a pro-repair immune environment into one that perpetuates catabolism.
From a translational standpoint, the most exciting contribution of Westermann et al. (2026) is the identification of metabolic enzymes and intermediates that could serve as therapeutic targets. By mapping the biochemical intersections between the cachectic and exercise states, the authors propose that compounds capable of mimicking the metabolic resolution phase of exercise could represent a new class of cachexia therapeutics — particularly relevant for patients too ill to exercise.
Cachexia and acute exercise share surprising molecular and metabolic similarities — including cytokine activation, mitochondrial stress, and altered amino acid catabolism — but differ fundamentally in the resolution of these stresses. Exercise achieves resolution through myokine-mediated immunomodulation and anabolic rebound; cachexia does not. This crosstalk reveals potential drug targets and supports the use of structured exercise as a therapeutic strategy in wasting conditions where feasible.
Synthesising the Evidence: A New Paradigm for Muscle Biology
1️⃣ Exercise Is No Longer a Lifestyle Recommendation — It Is Molecular Therapy
For decades, clinicians prescribed exercise as advice. Today, we must recognize it as intervention. Modern multi-omics research demonstrates that exercise activates hierarchically organized signaling networks — including AMPK, CaMKII, mTORC1, ROS-mediated transcriptional control, and mitochondrial biogenesis pathways. These are not vague wellness effects; they are targeted molecular cascades that reprogram cellular metabolism.
When we recommend exercise, we are prescribing controlled metabolic stress designed to induce adaptive remodeling.
2️⃣ Skeletal Muscle Is an Endocrine and Epigenetic Organ
The old model viewed skeletal muscle as a contractile engine. The new model reveals it as:
A myokine-secreting endocrine organ
A systemic immune modulator
An epigenetic memory reservoir
A metabolic command center
Contracting muscle releases signaling molecules that influence adipose tissue, liver metabolism, brain function, and immune polarization. The implications extend from insulin sensitivity to neuroprotection and inflammation control.
Medicine has not yet fully integrated this systemic paradigm.
3️⃣ Sex Is a Biological Variable — Not a Statistical Adjustment
Large-scale multi-omics integration has confirmed what physiology long suggested: men and women do not adapt identically at the molecular level. Lipid metabolism pathways, extracellular matrix remodeling, proteomic shifts, and glycolytic responses differ meaningfully between sexes.
This challenges one-size-fits-all exercise prescriptions — particularly in cardiac rehabilitation, oncology recovery, metabolic disease, and sarcopenia management.
Personalized exercise medicine must include sex-stratified design as standard practice.
4️⃣ Muscle Memory Is Epigenetic — and Clinically Exploitable
The concept of “muscle memory” extends beyond neuromuscular coordination. Prior training imprints durable epigenetic marks on mitochondrial biogenesis genes and metabolic regulators.
Clinically, this means:
A previously trained patient is not metabolically naïve
Detrained individuals retain molecular advantages
Rehabilitation can leverage prior conditioning history
In geriatrics, oncology, and post-surgical recovery, this insight reframes how we assess and reintroduce physical training.
5️⃣ Exercise and Disease Share Molecular Pathways
Perhaps the most provocative finding of recent research is the overlap between acute exercise stress and pathological states such as cachexia. Both involve inflammatory cytokine activation, altered amino acid metabolism, mitochondrial stress, and unfolded protein response signaling.
The difference is resolution.
Exercise induces transient stress followed by anabolic recovery. Disease sustains catabolism without resolution. Understanding this distinction opens doors to exercise mimetics and novel metabolic therapeutics for patients unable to train.
6️⃣ The Future of Medicine Will Be Exercise-Literate
Sequencing costs are falling. Multi-omics analytics are becoming clinically scalable. Within a decade, we may stratify exercise prescriptions based on transcriptomic responsiveness, mitochondrial phenotype, and inflammatory baseline signatures.
Exercise will no longer be generalized advice — it will be precision-dosed metabolic programming.
7️⃣ The Urgent Imperative
Medicine stands at a crossroads. We can continue treating exercise as supplementary lifestyle counseling, or we can integrate it into mainstream molecular therapeutics.
The science is no longer speculative.
Every workout is a biological signal — capable of reshaping metabolism, immunity, resilience, and aging trajectories.
Comparison of Signaling Hierarchies
Here is the breakdown of signaling hierarchies and how they translate from the microscopic level to the patient experience.
1. Endurance & HIIT: The Energy Factory Upgrade
Primary Molecular Driver: AMPK (AMP-activated protein kinase).
Scientific Perspective: High-intensity or sustained contraction creates energetic stress (ATP depletion). AMPK acts as the cell’s "fuel gauge," triggering mitochondrial biogenesis—the creation of new cellular power plants—to increase oxidative capacity and fatty acid oxidation.
This type of training is like upgrading your car’s engine. We are teaching your cells how to produce more energy and burn fuel more efficiently, which helps combat fatigue and improves your metabolic health."
2. Resistance Training: The Structural Armor
Primary Molecular Driver: mTORC1 (mechanistic target of rapamycin complex 1).
Scientific Perspective: Mechanical tension and amino acid availability activate the mTORC1 pathway, which is the master regulator of protein synthesis. This leads to myofibrillar remodeling and hypertrophy (muscle growth) by physically building new contractile units.
"Lifting weights sends a 'build' signal to your body. It’s not just about looks; it’s about reinforcing your 'structural armor.' We are literally adding new protein to your muscles to make you stronger and more resilient against injury."
3. Stretching & Yoga: The Architectural Reset
Primary Molecular Driver: Mechanotransduction (integrin-mediated signaling).
Scientific Perspective: Physical stretch pulls on the cell membrane, activating integrins that communicate with the Extracellular Matrix (ECM). This triggers fibroblasts to remodel collagen and fascia, improving tissue elasticity and reducing "stiffness" at a molecular level.
Stretching isn't just 'relaxing' the muscle; it’s a physical signal to the 'glue' that holds your body together. It tells your tissues to reorganize and become more supple, which helps your joints move through a full, pain-free range of motion."
4. The "Molecular Crosstalk": Why Variety Matters
Scientific Perspective: While these pathways are distinct, they are not isolated. For example, CaMKII links the electrical signal of a muscle contraction to the metabolic adaptations of both AMPK and mTORC1, ensuring the muscle adapts to the specific type of "stress" it receives.
Think of your body like a computer that needs different software updates. HIIT updates your battery life, resistance training updates your hardware, and stretching updates your flexibility. To stay fully 'functional,' you need a bit of each update."
Frequently Asked Questions (FAQs)
1. What does "multi-omics" mean, and why does it matter for exercise research? "Multi-omics" refers to the simultaneous measurement of multiple biological information layers — including the genome (DNA), transcriptome (RNA), proteome (proteins), metabolome (metabolites), and epigenome (chemical modifications on DNA and histones). Traditional studies typically examine only one layer at a time, which can miss interactions that only become visible when multiple layers are considered together. Jacques et al. (2025) used this integrated approach to reveal sex- and modality-specific signatures of exercise adaptation that single-omics studies had entirely missed, making multi-omics a critical methodological advance for the field.
2. Do men and women need to exercise differently based on this science? The evidence from Jacques et al. (2025) strongly suggests that the molecular responses to the same exercise dose differ meaningfully between biological sexes, with females showing greater transcriptional changes in lipid metabolism and extracellular matrix pathways, and males showing stronger glycolytic and contractile protein responses. Practically, this does not mean entirely different workout programmes, but it does justify sex-stratified research and potentially sex-tailored exercise prescriptions, particularly in therapeutic contexts such as rehabilitation or chronic disease management.
3. What is muscle memory at the molecular level? Beyond the motor-learning concept, molecular muscle memory refers to epigenetic modifications — changes to DNA methylation and histone marks — that persist in muscle cells long after a training programme ends. As described by Weidenhamer et al. (2025), these modifications "prime" mitochondrial biogenesis genes so that when exercise is resumed after a break, the metabolic response is faster and more robust. This is a key reason why previously trained individuals tend to regain fitness more quickly than beginners reaching the same level for the first time.
4. Is exercise genuinely beneficial for cancer patients experiencing cachexia? The evidence is growing. Westermann et al. (2026) explain that structured exercise can, in patients capable of performing it, partially counter the sustained inflammatory and catabolic signalling that drives cachexia by promoting transient myokine release and anabolic recovery windows. However, many cachectic patients are too ill to exercise, which is why the same research highlights the urgent need for pharmacological exercise mimetics that can replicate these metabolic benefits without requiring physical exertion.
5. What are myokines, and why are they important? Myokines are proteins secreted by skeletal muscle during contraction that act as signalling molecules on other tissues, including the immune system, the brain, the liver, and adipose tissue. Key exercise-induced myokines include IL-6, irisin, and brain-derived neurotrophic factor (BDNF). Westermann et al. (2026) describe how transient myokine elevation during exercise promotes anti-inflammatory immune polarisation — a benefit that is disrupted in cachexia. This is one key mechanism by which exercise exerts systemic health benefits far beyond the muscle itself.
6. Which type of exercise is molecularly "best" — HIIT, steady-state cardio, or resistance training? There is no single best modality; each produces a distinct and valuable molecular signature. According to Jacques et al. (2025), HIIT generates the broadest transcriptional response and the greatest mitochondrial pathway activation; resistance training uniquely drives large-scale proteome remodelling of sarcomeric (contractile) proteins; and MICT produces a more moderate but broad adaptive response. The optimal exercise modality depends on individual goals, biological sex, fitness level, and health status — reinforcing the concept of truly personalised exercise prescription.
7. How might this research eventually change clinical medicine? These studies collectively point toward several transformational applications. The multi-omics framework from Jacques et al. (2025) could inform sex-stratified exercise protocols in cardiac rehabilitation, oncology, and geriatric medicine. The muscle memory findings of Weidenhamer et al. (2025) could reshape return-to-play and post-surgical rehabilitation protocols by leveraging prior training history as a therapeutic asset. And the cachexia–exercise crosstalk mapped by Westermann et al. (2026) is already guiding drug discovery efforts aimed at developing exercise mimetics — compounds that could deliver muscle-protective metabolic benefits to patients physically unable to exercise.
Author’s Note
As a clinician in internal medicine and a lifelong student of metabolic science, I have watched the conversation around exercise evolve from simplistic calorie arithmetic to something far more profound. We now understand that when a patient lifts a weight, walks briskly, or performs interval training, they are not merely “burning energy.” They are activating ancient molecular circuits — stress-sensing kinases, mitochondrial regulators, epigenetic modifiers, and immune signaling networks that shape long-term physiology.
This article was written to bridge two worlds that too often remain separate: high-resolution molecular research and practical clinical medicine. The studies discussed here are not abstract laboratory findings; they are blueprints for how we might rethink prevention, rehabilitation, metabolic disease management, aging, and even cancer-associated muscle wasting. Exercise is emerging not as lifestyle advice, but as programmable biological therapy.
Importantly, this is an evolving field. Multi-omics technologies are advancing rapidly, and with them, our understanding of sex-specific adaptation, mitochondrial memory, and muscle–immune crosstalk continues to deepen. As with all scientific work, interpretations should remain open to refinement as new evidence emerges.
My goal is not to replace individualized medical guidance, but to elevate the discussion — to encourage clinicians, researchers, and informed readers to see skeletal muscle as the dynamic, system-wide regulator it truly is.
Exercise is not a peripheral intervention. It is one of the most powerful molecular tools we currently possess.
Medical Disclaimer
The information in this article, including the research findings, is for educational purposes only and does not constitute medical advice, diagnosis, or treatment. Before starting an exercise program, you must consult with a qualified healthcare professional, especially if you have existing health conditions (such as cardiovascular disease, uncontrolled hypertension, or advanced metabolic disease). Exercise carries inherent risks, and you assume full responsibility for your actions. This article does not establish a doctor-patient relationship.
Related Articles
No Time to Train? Science-Backed Workouts That Deliver | DR T S DIDWAL
Fasted Cardio: Is Training on an Empty Stomach a Fat Loss Myth? | DR T S DIDWAL
References
Jacques, M., Landen, S., Sharples, A. P., Garnham, A., Schittenhelm, R., Steele, J., Heikkinen, A., Sillanpää, E., Ollikainen, M., Broatch, J., Zarekookandeh, N., Hanson, O., Ekström, O., Asplund, O., Lamon, S., Alexander, S. E., Smith, C., Bauer, C., Woessner, M. N., … Eynon, N. (2025). Molecular landscape of sex- and modality-specific exercise adaptation in human skeletal muscle through large-scale multi-omics integration. Cell Reports, 44(6), 115750. https://doi.org/10.1016/j.celrep.2025.115750
Smith, J. A. B., Murach, K. A., Dyar, K. A., et al. (2023). Exercise metabolism and adaptation in skeletal muscle. Nature Reviews Molecular Cell Biology, 24, 607–632. https://doi.org/10.1038/s41580-023-00606-x
Weidenhamer, C. J., Huang, Y. H., Natua, S., Kalsotra, A., & Hernández-Saavedra, D. (2025). Muscle memory of exercise optimizes mitochondrial metabolism to support skeletal muscle growth. American Journal of Physiology–Cell Physiology, 329(4), C1239–C1255. https://doi.org/10.1152/ajpcell.00451.2025
Westermann, S., Bennühr, B. C., Fumo, A. R., Rohm, M., & Hiller, K. (2026). Muscle–immune metabolic crosstalk: Shared pathways in cachexia and exercise. Current Opinion in Biotechnology, 98, 103455. https://doi.org/10.1016/j.copbio.2026.103455