Is TSH More Than a Marker? Rethinking Its Role in Dyslipidemia and Cardiovascular Risk
Is TSH more than a thyroid marker? Discover how elevated TSH may directly influence dyslipidemia, LDL cholesterol, and cardiovascular risk
HEARTMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/14/202614 min read
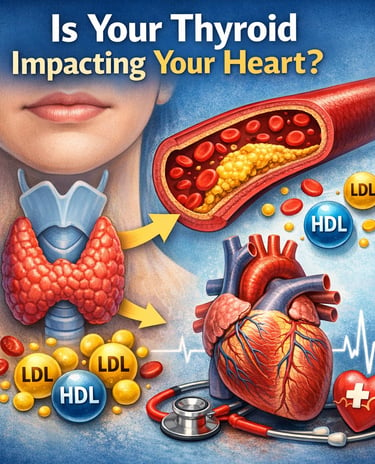
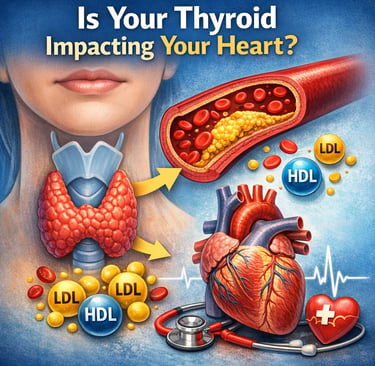
Hypothyroidism can raise cholesterol levels by slowing the body’s ability to clear LDL (“bad cholesterol”) and triglycerides from the bloodstream. Low thyroid hormone levels reduce liver LDL receptors and impair lipid metabolism, increasing the risk of dyslipidemia, atherosclerosis, and cardiovascular disease (Su et al., 2022; Gunvantbhai et al., 2025).
Most people think of the thyroid gland as a tiny organ that controls energy levels, body weight, and metabolism. But few realize that this small butterfly-shaped gland in the neck also plays a powerful role in regulating cholesterol and blood fats. In fact, disturbances in thyroid function are one of the most overlooked causes of abnormal cholesterol levels, medically known as dyslipidemia.
When the thyroid does not produce enough hormones—a condition called hypothyroidism—the body’s ability to process and clear cholesterol slows down. As a result, levels of low-density lipoprotein (LDL) cholesterol, often called “bad cholesterol,” begin to rise in the bloodstream. Triglycerides may also increase, while protective high-density lipoprotein (HDL) cholesterol can decline, creating a lipid pattern that significantly raises the risk of atherosclerosis, heart disease, and stroke (Su et al., 2022).
This connection between thyroid dysfunction and cholesterol metabolism is so strong that many cardiology and endocrinology guidelines recommend checking thyroid function in people with unexplained high cholesterol. Studies consistently show that patients with overt or even subclinical hypothyroidism often have higher total cholesterol and LDL levels compared with individuals whose thyroid function is normal (Gunvantbhai et al., 2025). Importantly, these changes can occur even when symptoms of thyroid disease—such as fatigue, weight gain, or cold intolerance—are mild or absent.
Recent research has also revealed that thyroid-stimulating hormone (TSH), the hormone commonly used to diagnose thyroid disorders, may directly influence lipid metabolism by acting on receptors in the liver and fat tissue (Wang et al., 2024). This means thyroid disorders may affect cholesterol levels not only through thyroid hormones themselves but also through TSH-mediated metabolic pathways.
Clinical pearls
1. The LDL Receptor "Clearance Clog"
Scientific Perspective: Hypothyroidism leads to a significant downregulation of hepatic LDL receptors. Even if the body is not overproducing cholesterol, the liver loses its "docking stations" to pull LDL out of the blood, resulting in a secondary hypercholesterolemia.
Think of your liver as a recycling center for fat. Thyroid hormones keep the doors open. When your thyroid is low, those doors lock shut—cholesterol isn't necessarily being created faster, but it’s piling up in your bloodstream because it has nowhere to go.
2. TSH: More Than Just a "Thermostat"
Scientific Perspective: Beyond its role in the pituitary-thyroid axis, TSH is an independent metabolic signaling molecule. TSH receptors are located in the liver and adipose tissue; elevated TSH can directly stimulate the creation of fats (lipogenesis) even before T3 and T4 levels drop significantly.
Most people think TSH is just a messenger that tells the thyroid to wake up. We now know TSH can act on its own to tell your body to store fat. This is why even "mild" thyroid issues can cause your cholesterol numbers to jump.
3. The "Bile Acid" Exit Route
Scientific Perspective: Thyroid hormones activate the CYP7A1 enzyme, which is the rate-limiting step in converting cholesterol into bile acids for excretion. Hypothyroidism suppresses this enzyme, effectively blocking the body's primary "exit ramp" for excess cholesterol.
Your body gets rid of extra cholesterol by turning it into bile (which helps you digest food). A slow thyroid turns off the tap to that drain, forcing the body to keep cholesterol circulating in the blood instead of flushing it out.
4. The Subclinical "Silent Risk"
Scientific Perspective: Subclinical hypothyroidism (elevated TSH with normal FT4) is often associated with a pro-atherogenic lipid profile. Research suggests that the threshold for treating high cholesterol may need to be adjusted in these patients to prevent early-stage cardiovascular damage.
You don’t need a "failed" thyroid test to be at risk. Even if your hormone levels are technically in the "normal range," if your TSH is creeping up, your heart and arteries might already be feeling the strain of rising cholesterol levels.
5. Post-Treatment "Residual Risk"
Scientific Perspective: Normalizing TSH with levothyroxine does not always fully resolve the molecular lipid abnormalities identified in lipidomics. This suggests that some "atherogenic" (heart-damaging) lipid species may remain dysregulated even when standard blood tests look perfect.
Getting your thyroid "numbers" back to normal is the first step, but it’s not the finish line. Because thyroid issues change the quality of your cholesterol, not just the quantity, you should still keep a close eye on your heart health even after your thyroid medication is dialled in.
Hypothyroidism and High Cholesterol: The Hidden Thyroid–Heart Connection
The relationship between thyroid function and lipid metabolism is one of endocrinology's most well-established and clinically significant connections — and it is still revealing new complexity. The six studies reviewed in this article collectively demonstrate that hypothyroidism drives dyslipidemia through multiple converging biochemical pathways, that the severity of lipid disruption is modulated by age and population factors, that TSH itself is an active metabolic regulator rather than a passive biomarker, and that molecular-level lipid changes may persist even after conventional treatment.
For patients living with thyroid conditions, this research highlights the importance of comprehensive metabolic monitoring — not just tracking TSH and thyroid hormones, but also regularly assessing the full lipid profile as part of cardiovascular risk management. For clinicians, it calls for an integrated approach that considers thyroid and cardiovascular health as inseparable components of the same metabolic system.
How hypothyroidism drives dyslipidemia
Hypothyroidism disrupts several key pathways of lipid metabolism, leading to characteristic changes in the cholesterol profile. Low thyroid hormone levels reduce the expression of hepatic LDL receptors, impairing the liver’s ability to remove LDL (“bad cholesterol”) from the bloodstream and causing LDL levels to rise. At the same time, reduced activation of the CYP7A1 enzyme limits the conversion of cholesterol into bile acids, blocking one of the body’s primary routes for cholesterol elimination and increasing total cholesterol. Hypothyroidism also suppresses lipoprotein lipase (LPL) activity, slowing the breakdown of triglyceride-rich lipoproteins such as VLDL and leading to elevated triglycerides. In addition, thyroid hormone deficiency impairs reverse cholesterol transport, reducing the efficiency of HDL in carrying excess cholesterol from peripheral tissues back to the liver for disposal. Together, these mechanisms explain why hypothyroidism is strongly associated with high LDL cholesterol, elevated triglycerides, reduced HDL, and an increased risk of dyslipidemia and cardiovascular disease.
Study 1: A Comparative Perspective from Ethiopia
Kebamo et al. (2025) offer a valuable perspective from a low-to-middle-income country (LMIC) context. While much of the existing literature on thyroid dysfunction and dyslipidemia originates from high-income settings, this study sheds light on how the thyroid–lipid relationship manifests in Ethiopian patients — a population with distinct dietary patterns, healthcare access dynamics, and baseline metabolic profiles.
The study compared serum lipid levels — including total cholesterol (TC), LDL cholesterol, HDL cholesterol, and triglycerides (TG) — across patients with hypothyroidism, hyperthyroidism, and euthyroid (normal thyroid function) controls. Consistent with global findings, hypothyroid patients demonstrated significantly higher TC and LDL levels, while hyperthyroid patients tended to exhibit lower lipid concentrations. The study also highlighted that dyslipidemia in this population is often underdiagnosed and undertreated, underscoring the importance of routine lipid screening in thyroid patients regardless of geographic or economic setting.
Key Takeaway: Thyroid dysfunction is associated with measurable lipid abnormalities even in low-resource settings like Ethiopia, reinforcing the universal importance of lipid screening across all thyroid patients globally, not just in high-income nations.
Study 2: Lipid Profile Abnormalities in Hypothyroidism — Indian Clinical Evidence
Gunvantbhai et al.(2025) examined the association between thyroid hormone concentrations and lipid profile abnormalities specifically in hypothyroid patients. Published in the International Journal of Current Pharmaceutical Review and Research, the study evaluated serum levels of free thyroxine (FT4), free triiodothyronine (FT3), and TSH in correlation with TC, LDL, HDL, and TG in a clinical cohort.
The findings reinforced the well-established inverse relationship between thyroid hormone levels and atherogenic lipids. As thyroid hormone concentrations fell, TC and LDL rose — a pattern driven by reduced hepatic expression of LDL receptors. When fewer LDL receptors are present on liver cells, circulating LDL cannot be efficiently cleared from the bloodstream, leading to hypercholesterolemia. The study also found that TSH levels had a significant positive correlation with TC and LDL, suggesting TSH itself may contribute directly to lipid dysregulation rather than merely serving as a marker of thyroid deficiency.
They emphasized that even subclinical hypothyroidism — a condition where TSH is mildly elevated but thyroid hormones remain within the normal range — may produce enough lipid disruption to warrant clinical attention. This finding has direct implications for cardiovascular risk stratification and treatment thresholds.
🔑 Key Takeaway: Even mild or subclinical hypothyroidism can elevate cholesterol and LDL to clinically meaningful levels. TSH elevation alone — without overt hormone deficiency — may be an independent signal for lipid dysregulation, warranting proactive cardiovascular monitoring.
Study 3: Mechanistic Pathways Behind Hyperlipidemia in Hypothyroidism
While the clinical associations between hypothyroidism and dyslipidemia are well-documented, the biochemical mechanisms behind them were comprehensively reviewed by Su et al. (2022) in Clinica Chimica Acta. This review is essential reading for understanding not just what happens when the thyroid fails, but why.
Thyroid hormones — primarily triiodothyronine (T3) — regulate several critical steps in lipid metabolism. First, T3 upregulates the expression of LDL receptors on hepatocytes, enabling the liver to clear LDL from the blood. In hypothyroidism, receptor expression drops, and LDL accumulates. Second, thyroid hormones activate cholesterol 7-alpha-hydroxylase (CYP7A1), the rate-limiting enzyme in bile acid synthesis. Reduced CYP7A1 activity means less cholesterol is converted into bile acids and excreted, further raising plasma cholesterol. Third, lipoprotein lipase (LPL) activity — responsible for hydrolyzing triglycerides from very-low-density lipoproteins (VLDL) — is also suppressed in hypothyroidism, contributing to hypertriglyceridemia.
Su et al. (2022) also discussed the emerging role of thyroid hormone signaling in reverse cholesterol transport, the process by which HDL retrieves excess cholesterol from peripheral tissues and returns it to the liver. Disruptions in this pathway in hypothyroid states help explain why HDL cholesterol levels often fall, compounding the overall atherogenic lipid profile.
🔑 Key Takeaway: The thyroid–lipid relationship is mechanistically rich: hypothyroidism impairs LDL receptor expression, suppresses bile acid synthesis, reduces lipoprotein lipase activity, and disrupts reverse cholesterol transport — creating a perfect metabolic storm for cardiovascular risk.
Study 4: TSH and Lipids in Elderly Hypothyroid Patients
Age is a critical modifier in thyroid-lipid research. Fotedar et al. (2025), publishing in the European Journal of Cardiovascular Medicine, focused specifically on geriatric patients — a population in which both hypothyroidism and dyslipidemia are more prevalent, and in which cardiovascular risk is already elevated. Their cross-sectional study assessed lipid profile alterations and their correlation with TSH levels in older adults with confirmed hypothyroidism.
The study found a strong positive correlation between TSH concentrations and TC and LDL levels in the geriatric cohort, consistent with findings in younger populations. However, the magnitude of lipid abnormalities was notably more pronounced in the elderly, highlighting that age-related metabolic changes — including reduced hepatic clearance, altered hormone receptor sensitivity, and lower basal metabolic rate — amplify the lipid-raising effects of hypothyroidism.
The authors emphasized that geriatric hypothyroid patients represent a particularly high-risk group for cardiovascular morbidity. They called for lower thresholds for lipid screening and thyroid hormone replacement therapy in older adults, and noted that the combined burden of aging-related lipid changes and hypothyroid-driven dyslipidemia can significantly shorten healthy life expectancy if left unaddressed.
🔑 Key Takeaway: Elderly hypothyroid patients face compounded metabolic vulnerabilities — the lipid-raising effects of thyroid deficiency are amplified by age-related metabolic decline, making this group a high-priority population for early and proactive cardiovascular intervention.
Study 5: Lipidomics and the Evolving Science of Hypothyroid Dyslipidemia
The field of lipidomics — the comprehensive study of lipid species and their biological functions — is opening new frontiers in thyroid research. Jonklaas (2024), writing in the journal Endocrine, provides a thought-provoking review of what lipidomics is beginning to reveal about hypothyroidism and lipid metabolism that traditional lipid panels (TC, LDL, HDL, TG) simply cannot capture.
Standard lipid testing measures bulk lipid concentrations, but lipidomics identifies specific lipid species — including distinct subtypes of phospholipids, sphingolipids, and fatty acids — that may be dysregulated independently of standard lipid markers. Jonklaas (2024) reviews emerging evidence suggesting that hypothyroidism alters the composition of lipoprotein particles in ways that may increase their atherogenicity even when conventional lipid numbers appear only mildly elevated.
This has significant implications for cardiovascular risk assessment. A patient with moderately elevated LDL may have a much higher actual cardiovascular risk if the specific composition of those LDL particles is particularly atherogenic — something only lipidomics can reveal. Jonklaas also explored how thyroid hormone replacement therapy (levothyroxine) affects lipidomic profiles, finding that normalization of thyroid hormone levels does not always fully resolve lipid abnormalities at the molecular species level, pointing to residual cardiovascular risk even in adequately treated patients.
🔑 Key Takeaway: Lipidomics reveals that hypothyroidism alters not just lipid quantities but lipid quality and composition at the molecular level — and that standard levothyroxine therapy may not fully resolve these atherogenic changes, suggesting a need for more nuanced post-treatment monitoring.
Study 6: TSH as an Independent Regulator of Lipid Metabolism and Brain–Body Communication
Perhaps the most conceptually ambitious of the six studies, Wang et al. (2024), published in Neurobiology of Disease, explores TSH not merely as a pituitary hormone that stimulates thyroid function, but as an independent metabolic regulator with direct effects on lipid metabolism — effects that extend beyond the thyroid gland to influence communication between the body and the brain.
The research demonstrates that TSH receptors are expressed not only in thyroid tissue but also in adipose tissue, liver, and the brain. This means that elevated TSH levels — as seen in hypothyroidism — can directly stimulate lipid synthesis in peripheral tissues and potentially alter neurological lipid signaling pathways. The study examined how TSH-mediated lipid dysregulation contributes to neuroinflammation, cognitive changes, and mood disturbances sometimes observed in hypothyroid patients — symptoms traditionally attributed solely to thyroid hormone deficiency.
Wang et al. (2024) present a model in which the brain is not a passive recipient of peripheral metabolic dysfunction, but an active participant — both influencing and being influenced by TSH-driven lipid changes. This body–brain communication framework suggests that the neuropsychiatric symptoms of hypothyroidism may be at least partly mediated through lipid metabolic pathways regulated by TSH itself, independent of T3 and T4 levels.
🔑 Key Takeaway: TSH is far more than a thyroid-stimulating signal — it acts as an independent lipid metabolic regulator with receptors in the liver, fat tissue, and brain. Elevated TSH may directly drive both peripheral dyslipidemia and neurological consequences, redefining how we understand hypothyroid-related cognitive and mood symptoms.
Synthesising the Evidence: A Unified View of Thyroid–Lipid Interactions
1. The Thyroid Is a Central Regulator of Lipid Metabolism
Thyroid hormones—particularly triiodothyronine (T3)—control several critical enzymes and receptors involved in lipid metabolism. They regulate hepatic LDL receptor expression, stimulate cholesterol conversion into bile acids, and influence lipoprotein lipase activity, which helps break down triglycerides. When thyroid hormone levels fall, these processes slow down, allowing cholesterol and triglycerides to accumulate in the bloodstream (Su et al., 2022).
2. Hypothyroidism Is a Common Secondary Cause of High Cholesterol
Clinical studies consistently show that patients with hypothyroidism exhibit elevated total cholesterol, LDL cholesterol, and triglycerides, often accompanied by reduced HDL cholesterol. This lipid pattern is strongly associated with atherosclerosis and cardiovascular disease. Importantly, even subclinical hypothyroidism—where TSH is elevated, but thyroid hormone levels remain within the normal range—can still contribute to significant lipid abnormalities (Gunvantbhai et al., 2025).
3. Thyroid-Stimulating Hormone May Directly Influence Lipid Biology
Emerging evidence suggests that thyroid-stimulating hormone (TSH) may play a more active metabolic role than previously recognized. TSH receptors have been identified in liver tissue, adipose tissue, and even the brain, indicating that elevated TSH may directly promote lipid synthesis and metabolic dysregulation independent of thyroid hormone levels (Wang et al., 2024).
4. Age Amplifies the Cardiovascular Impact of Thyroid Dysfunction
Older adults represent a particularly vulnerable population. Studies show that the relationship between TSH levels and elevated LDL cholesterol becomes stronger with age, suggesting that thyroid-related lipid abnormalities may contribute disproportionately to cardiovascular risk in elderly patients (Fotedar et al., 2025).
5. Thyroid Treatment Improves—but May Not Fully Normalize—Lipid Metabolism
Restoring euthyroid status with levothyroxine therapy often leads to significant improvements in cholesterol levels. However, newer lipidomic research suggests that subtle changes in lipoprotein composition may persist even after thyroid hormones are normalized, indicating a potential residual cardiovascular risk that warrants ongoing lipid monitoring (Jonklaas, 2024).
The evidence is clear: thyroid function and cardiovascular health are metabolically intertwined. For clinicians, unexplained dyslipidemia should prompt thyroid evaluation, while patients with thyroid disorders should routinely undergo lipid assessment. Recognizing this connection allows earlier intervention and more comprehensive strategies for preventing long-term cardiovascular disease.
Frequently Asked Questions (FAQs)
Q: Does hypothyroidism always cause high cholesterol?
A: Not always — but elevated cholesterol is one of the most common manifestations of hypothyroidism, particularly overtly high TSH states. Even subclinical hypothyroidism (mildly elevated TSH with normal T3/T4) can raise LDL and total cholesterol to clinically meaningful levels, as demonstrated by Gunvantbhai et al. (2025) and Fotedar et al. (2025). However, the degree of lipid elevation varies by individual, disease severity, age, and other metabolic factors.
Q: Will treating hypothyroidism with levothyroxine normalize my cholesterol?
A: Thyroid hormone replacement often significantly improves lipid profiles, and many patients see LDL and total cholesterol fall substantially after achieving euthyroid status. However, Jonklaas (2024) presents lipidomic evidence suggesting that even optimally treated patients may retain subtle lipid abnormalities at the molecular species level. Regular lipid monitoring remains important even after thyroid hormone normalization.
Q: What lipid changes are associated with hyperthyroidism?
A: Hyperthyroidism generally produces the opposite lipid pattern to hypothyroidism — lower total cholesterol, lower LDL, and lower triglycerides, with variable effects on HDL. The Kebamo et al. (2025) comparative study from Ethiopia confirmed these directionally opposite lipid effects across hypothyroid and hyperthyroid patient groups. However, rapid cholesterol lowering in hyperthyroidism can mask underlying cardiovascular risks, and treatment-induced shifts in thyroid status require monitoring.
Q: Is TSH itself harmful to lipid levels, or is it just a marker of low thyroid hormones?
A: Emerging research, particularly the work of Wang et al. (2024), strongly suggests TSH plays an independent causal role in lipid dysregulation. TSH receptors expressed in the liver and adipose tissue allow TSH to directly stimulate lipid synthesis pathways. This means elevated TSH — even with marginally normal thyroid hormone levels — may independently worsen the lipid profile, rather than merely reflecting the consequences of hormone deficiency.
Q: Should elderly patients with hypothyroidism be treated more aggressively for dyslipidemia?
A: The evidence from Fotedar et al. (2025) supports heightened vigilance in geriatric hypothyroid patients. Age-related metabolic changes amplify the lipid-raising effects of hypothyroidism, and the combined cardiovascular risk burden in this group is substantially higher than in younger patients. Lower screening thresholds, more proactive lipid management, and careful thyroid replacement titration are all supported by the available evidence in this population.
Q: Can the mechanisms linking thyroid function and lipids explain brain-related symptoms of hypothyroidism?
A: Yes — and this is one of the most exciting new areas of research. Wang et al. (2024) demonstrated that TSH receptors are expressed in the brain, and that TSH-driven lipid metabolic changes may contribute to neuroinflammation and the cognitive and mood symptoms frequently reported by hypothyroid patients. This brain–body communication framework suggests that the neuropsychiatric burden of hypothyroidism is not solely due to thyroid hormone deficiency, but also to TSH-mediated disruptions in lipid signaling.
Q: Are the thyroid–lipid findings consistent across different populations globally?
A: Remarkably, yes. The studies reviewed here span Ethiopia, India, China, the United States, and Europe — and the core finding that hypothyroidism raises atherogenic lipids while hyperthyroidism lowers them is consistent across all settings. The Kebamo et al. (2025) Ethiopian study is particularly important in confirming that these mechanisms are universal and not limited to Western or high-income populations. Dietary patterns, genetic backgrounds, and healthcare access differ significantly across these regions, yet the thyroid–lipid axis remains robustly consistent.
Author’s Note
The relationship between thyroid function and lipid metabolism is one of the most fascinating examples of how endocrine regulation influences whole-body metabolic health. While thyroid disorders are commonly associated with symptoms such as fatigue, weight changes, or temperature intolerance, their effects on cholesterol metabolism and cardiovascular risk often receive less attention in everyday clinical discussions.
The purpose of this article is to bring together recent research from multiple regions of the world and translate it into a clear, clinically meaningful narrative. The studies reviewed here highlight that the thyroid–lipid axis is not a simple cause-and-effect relationship, but rather a complex network involving thyroid hormones, thyroid-stimulating hormone (TSH), liver metabolism, lipid transport pathways, and even emerging molecular insights from lipidomics research.
For patients, the key message is that abnormal cholesterol levels may sometimes have an endocrine cause. Identifying and treating thyroid dysfunction can significantly improve lipid profiles and may reduce long-term cardiovascular risk. For clinicians and healthcare professionals, the growing body of evidence reinforces the importance of viewing thyroid disease within a broader metabolic framework that includes cardiovascular and neurological health.
Medical science continues to evolve, and newer tools such as lipidomic profiling and molecular receptor studies are expanding our understanding of how thyroid signaling affects lipid metabolism at both systemic and cellular levels. As research progresses, these insights may help refine treatment strategies and improve risk prediction for patients with thyroid disorders.
This article is intended for educational purposes and reflects current scientific knowledge based on peer-reviewed studies. Patients should always consult their healthcare provider for personalized medical advice regarding thyroid conditions, cholesterol management, and cardiovascular risk.
References
Fotedar, S., Soothwal, S., Singh, S., Thakur, D. C., & Bansal, A. (2025). Lipid profile alterations and their correlation with thyroid-stimulating hormone levels in geriatric hypothyroid patients: A cross-sectional study. European Journal of Cardiovascular Medicine, 15(3), 728–732. https://doi.org/10.5083/ejcm/25-03-125
Gunvantbhai, P. A., Pirani, R., & Brahmbhatt, M. (2025). Association of thyroid hormones with lipid profile abnormalities in hypothyroidism. International Journal of Current Pharmaceutical Review and Research, 17(10), 487–491.
Jonklaas, J. (2024). Hypothyroidism, lipids, and lipidomics. Endocrine, 84(2), 293–300. https://doi.org/10.1007/s12020-023-03420-9
Kebamo, T. E., Tantu, A., Solomon, Y., et al. (2025). A comparative study on serum lipid levels in patients with thyroid dysfunction: A single-center experience in Ethiopia. BMC Endocrine Disorders, 25, 47. https://doi.org/10.1186/s12902-025-01851-1
Su, X., Peng, H., Chen, X., Wu, X., & Wang, B. (2022). Hyperlipidemia and hypothyroidism. Clinica Chimica Acta, 527, 61–70. https://doi.org/10.1016/j.cca.2022.01.006
Wang, X., Wu, Z., Liu, Y., Wu, C., Jiang, J., Hashimoto, K., & Zhou, X. (2024). The role of thyroid-stimulating hormone in regulating lipid metabolism: Implications for body–brain communication. Neurobiology of Disease, 201, Article 106658. https://doi.org/10.1016/j.nbd.2024.106658